Shoot Rot of Spikenard Caused by Rhizoctonia solani AG-2-1
Article information
Trans Abstract
In January 2021, unusual outbreak of shoot rot symptoms was observed in young spikenard (Aralia cordata) plants growing in vinyl greenhouses located in Chuncheon and Yanggu, Gangwon Province, Korea. The symptoms initially appeared on young shoots of the plants at or above the soil surface level. Later, the infected shoots wholly rotted and blighted. The incidence of diseased plants in the vinyl greenhouses investigated ranged from 5% to 30%. Eight isolates of Rhizoctonia sp. were obtained from shoot lesions of the diseased plants. All the isolates were identified as Rhizoctonia solani AG-2-1 based on the morphological characteristics and anastomosis test. Three isolates of R. solani AG-2-1 were tested for pathogenicity on young shoots of spikenard plants using artificial inoculation. All the tested isolates induced shoot rot symptoms on the inoculated plants. The symptoms were similar to those observed in spikenard plants from the vinyl greenhouses investigated. This is the first report of R. solani AG-2-1 causing shoot rot in spikenard.
Spikenard (Aralia cordata) belongs to the family Araliaceae, and its native range is China, Korea, Japan, Sakhalin, and Taiwan (Plants of the World Online, 2022). The plant has been cultivated in Korea, and young shoots of the plant are used as a vegetable. In January 2021, unusual outbreak of shoot rot symptoms was observed in young spikenard plants growing in vinyl greenhouses located in Chuncheon and Yanggu, Gangwon Province, Korea. The symptoms initially appeared on young shoots of the plants at or above the soil surface level. The infected shoots turned black and rotted (Fig. 1A–C), and severely diseased shoots wholly rotted and blighted. Eight vinyl greenhouses were investigated for the disease incidence in the two locations. One hundred plants in a vinyl greenhouse were investigated for the disease incidence. The disease occurred on the plants in five out of the vinyl greenhouses investigated, and incidence of diseased plants ranged from 5% to 30%.

Shoot rot of spikenard plants. (A–C) Symptoms observed in the vinyl greenhouse investigated. (D, E) Symptoms induced by artificial inoculation tests with Rhizoctonia solani AG-2-1 isolates. (F) Non-inoculated plants (control).
Diseased shoots of spikenard plants were collected from the vinyl greenhouses, and fungal pathogens were isolated from the shoot lesions. The 3–5 mm-long lesion pieces cut from the diseased shoots were surface-sterilized with 1% sodium hypochlorite solution for 1 min and plated on 2% water agar. The fungal mycelia growing from the lesion pieces were transferred to potato dextrose agar (PDA) slants after incubating the plates at 25° C for 2 days. Eight isolates of Rhizoctonia sp. were obtained from the shoot lesions and examined for their morphological characteristics using a compound microscope (Nikon Eclipse Ci-L, Tokyo, Japan). Branching near the distal septum of cells in young vegetative hyphae and constriction of hyphae and formation of septa at short distance from the point of hyphal branches were observed. In addition, the isolates did not produce conidia and clamp connections on hyphae. All the isolates were identified as Rhizoctonia solani Kühn based on the morphological characteristics as per descriptions from previous studies (Parmeter and Whitney, 1970; Sneh et al., 1991).
The R. solani isolates were tested for identification of anastomosis groups using tester isolates of R. solani (AG-1 through AG-5) as previously described (Kim et al., 1994, 2020). All the tested isolates were classified as R. solani AG-2-1. Anastomosis reactions between the tested isolate and the tester isolate of R. solani AG-2-1 are shown in Fig. 2A. The fused cells of the tested isolate and the tester isolate showing killing reactions were stained blue with 0.1% aniline blue solution. The colony of the isolates cultured on PDA displayed brown to reddish brown mycelia with concentric zones (Fig. 2B). Sclerotia were mostly formed in the concentric zones on the medium.
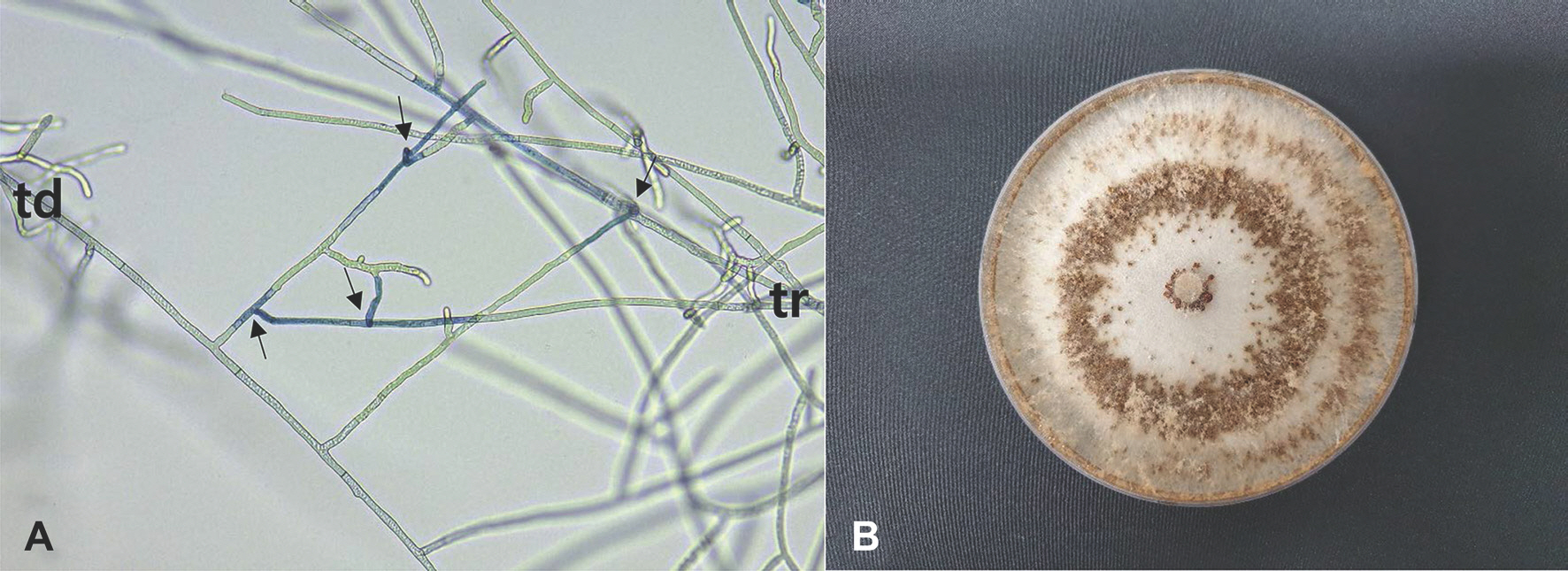
Anastomosis test of Rhizoctonia solani isolate from spikenard and cultural appearance of the isolate. (A) Anastomosis reactions between the tested isolate (td) and the tester isolate (tr) of R. solani AG-2-1. The arrows indicate points of hyphal anastomosis. (B) A colony of R. solani AG-2-1 isolate grown on potato dextrose agar at 25° C for 10 days.
Three isolates of R. solani AG-2-1 were tested for pathogenicity on young shoots of overwintered spikenard roots using artificial inoculation. Spikenard roots were planted in 1/2,000a Wagner pots (height, 29.7 cm; upper diameter, 25.6 cm; lower diameter, 23.4 cm), and cultivated in a vinyl greenhouse. Mycelial disks of 6 mm in diameter were cut from the margins of actively growing cultures of each isolate on PDA, and placed on shoots 1–2 cm above the soil surface of spikenard plants grown in the Wagner pots. PDA disks of 6 mm in diameter were used for the control plants. The inoculated plant pots were put in plastic bags of which inside was wetted with sterile distilled water. They were placed indoors at room temperature (22–26°C) for 3 days. Thereafter, the plastic bags were removed from the inoculated plant pots. Pathogenicity of the isolates was rated based on the degree of shoot rot symptoms 5 days after inoculation. The inoculation test was conducted in triplicate.
All the tested isolates of R. solani AG-2-1 induced shoot rot symptoms in the inoculated plants (Fig. 1D, E), but no symptoms occurred in the control plants (Fig. 1F). The symptoms induced by the artificial inoculation were similar to those observed in spikenard plants from the vinyl greenhouses investigated. The inoculated isolates were re-isolated from the lesions.
Anastomosis groups of R. solani have different genetic and pathological characteristics (Sneh et al., 1991). It has been reported that R. solani AG-2-1 causes damping-off, bottom rot, root rot, crown rot, bud rot, and leaf blight in various crops (Kim, 1996). However, there has been no report on shoot rot of spikenard caused by R. solani AG-2-1. This is the first report of R. solani AG-2-1 causing shoot rot in spikenard.
Notes
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Acknowledgments
This study was supported by a research grant (PJ014507012020) from the Rural Development Administration, Korea and a research grant (LP0018752021) from the Wild Vegetable Research Institute, Gangwondo Agricultural Research and Extension Services, Korea.
