Current Status and Future Prospects of White Root Rot Management in Pear Orchards: A Review
Article information
Abstract
The current social demand for organic, sustainable, and eco-friendly approaches for farming, while ensuring the health and productivity of crops is increasing rapidly. Biocontrol agents are applied to crops to ensure biological control of plant pathogens. Research on the biological control of white root rot disease caused by a soil-borne pathogen, Rosellinia necatrix, is limited in pears compared to that in apple and avocado. This pathogenic fungus has an extensive host range, and symptoms of this disease include rotting of roots, yellowing and falling of leaves, wilting, and finally tree death. The severity of the disease caused by R. necatrix, makes it the most harmful fungal pathogen infecting the economical fruit tree species, such as pears, and is one of the main limiting factors in pear farming, with devastating effects on plant health and yield. In addition to agronomic and cultural practices, growers use chemical treatments to control the disease. However, rising public concern about environmental pollution and harmful effects of chemicals in humans and animals has facilitated the search for novel and environmentally friendly disease control methods. This review will briefly summarize the current status of biocontrol agents, ecofriendly methods, and possible approaches to control disease in pear orchards.
Introduction
Rosellinia necatrix Berl. ex Prill. (anamorph Dematophora necatrix R. Hartig), a necrotrophic phytopathogenic fungus that threatens myriad of species, is reported in different countries worldwide (Kulshrestha et al., 2014; Narváez et al., 2020). Its host range comprising more than 400 plant species has been listed by the USDA-ARS (United States Department of Agriculture, Agricultural Research Service, http://nt.arsgrin.gov/fungaldatabases/index.cfm), which is frequently updated because several new hosts are continuously identified and added to the database. The fungus R. necatrix, which belongs to the Xylariaceae family, is a soil-borne fungus that causes white root rot, which seriously damages the roots of economically important plants worldwide (Kulshrestha et al., 2014). R. necatrix infects both the surfaces of roots and the tissue under the bark in a broad range of economically important crops, such as cotton, nuts, apples, cherries, avocados, and pears (Eguchi et al., 2008; Lee et al., 2020; Pérez-Jiménez, 2006; Ten Hoopen and Krauss, 2006). R. necatrix-mediated disease cause physically devastating symptoms in fruit trees, which makes it one of the most significant agricultural problems in the world (Kim et al., 2019). White root rot disease results in varying degrees of canopy decline, followed by leaf drop, wilting, and eventually the death of infected plants (Kim et al., 2019). Moreover, the pathogen can survive and remain active on the residues of susceptible crops such as olives, grapevines, and almonds for many years (Cazorla et al., 2006). R. necatrix can survive as saprophytes on dead roots and other plant debris in the soil, although fresh vegetable debris rich in cellulose is required for its continued survival. Under laboratory conditions, the pathogen survived on pear branches for a period of 18 months and died when the cellulose content decreased to 50% of the initial content (Araki, 1967). R. necatrix spreads in soil via mycelial growth, which proliferates and aggregates on the roots of host plants, and the pathogen may disperse in the soil via direct root-to-root contact between host plants and healthy plants (Pliego et al. 2009).
Currently, R. necatrix is widely dispersed in different hosts and has been detected in temperate, tropical, and subtropical regions in all five continents. It has been detected in countries such as China, South Korea, India, Iran, Pakistan, Australia, Mexico, Dominican Republic, Colombia, Brazil, the Democratic Republic of Congo, and Ethiopia (CABI, 2020). In Korea, this disease is posing a serious problem in pear and apple cultivation. The disease prevalent in both nurseries and orchards is destructive, resulting in substantial loss of yield and quality for apple and pear growers in Korea (Lee et al., 1995, 2006, 2016, 2020).
Pears belong to the genus Pyrus (Rosaceae, Pyrinae), and are commercially important fruit trees. They are one of the oldest plants cultivated by humans. Pear production worldwide is second to that of apples, and the world production for 2020/21 is anticipated to reach over 1.0 million tons to 22.2 million tons. In 2019, 23.9 million tons of pear fruit were produced in over 80 countries (FAOSTAT, http://faostat.fao.org/). Fresh pear fruit is consumed globally and is also found in processed products such as drinks, candy, preserved fruits, and jam. However, irrespective of its health benefits and economic value, the production levels and number of pear orchards have been progressively decreasing. A major reason for low production is the prevalence of diseases that appear at various growth stages and are difficult to manage. White root rot is a major problem that causes tree mortality. This disease is rather destructive as it eradicates the established orchards and causes considerable loss in the nursery. Root rot diseases infect deep in soil, and their management using chemicals is cumbersome and results in pollution. Therefore, the use of eco-friendly methods or bio-agents is a good alternative for managing such diseases. The use of biocontrol agents has received the most attention with respect to ecofriendly disease management, and necessitates implementation in pear orchards. In this review, we provide an overview of biocontrol agents, and available ecofriendly methods that can be used to control disease in pear orchards.
Control of White Root Rot Caused by Rosellinia necatrix
Controlling white root rot in the field has been proven difficult because of its tolerance and survival under drought conditions, tolerance to a wide range of soil pH, wide host range, and resistance to common fungicides (Khan, 1959). Therefore, to control this pathogen in the field, methods should focus on reducing the impact and spread of disease, which avoids chemicals and utilizes biological control agents (BCA), thereby implementing eco-friendly methods.
Biological Control
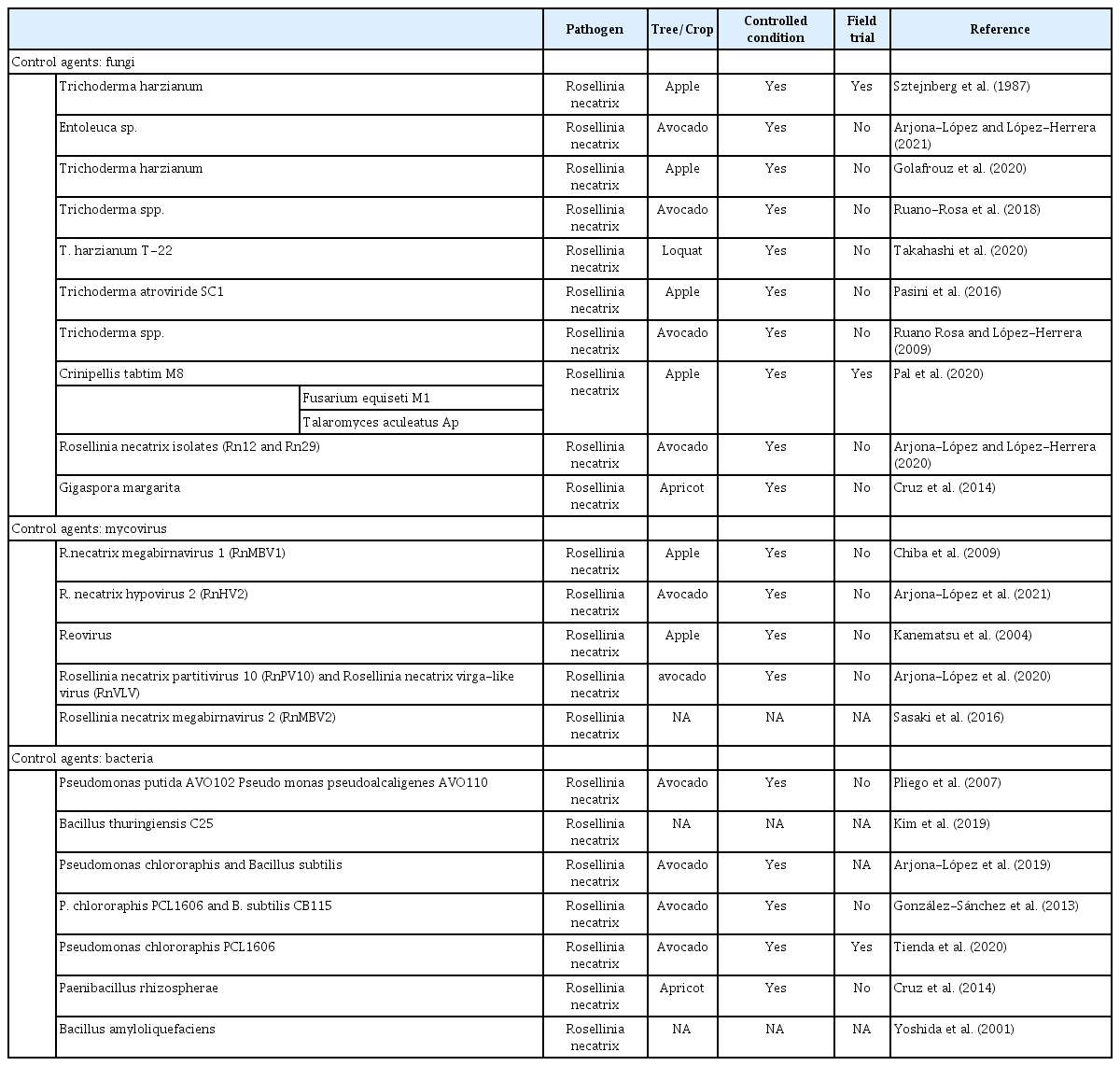
Numerous studies have been published on biological control of white root rot disease caused by R. necatrix by exploiting BCA, which has received considerable attention. Relatively few studies have focused on disease control in the field. Traditionally, growers use chemical fungicides to reduce the disease incidence in orchards. However, due to environmental concern, application of different BCA such as fungal antagonist, antagonistic bacteria, and mycoviruses are effective tools against white root rot caused by R. necatrix (Table 1).
Fungal Antagonist
Various fungal species have been used as biocontrol agents against R. necatrix (Table 1). These antagonistic fungi reduce mycelial growth in vitro and decrease disease symptoms caused by R. necatrix in different fruit tree crops such as apples, avocados, and apricots (Cruz et al., 2014; Pal et al., 2020; Ruano-Rosa et al., 2018). Among fungal genera investigated as biocontrol agents, Trichoderma has received considerable attention and has been successfully implemented as a BCA against R. necatrix in apple orchards (Golafrouz et al., 2020; Pasini et al., 2016; Sztejnberg et al., 1987) and avocado plants (Ruano Rosa and López-Herrera, 2009; Ruano-Rosa et al., 2018). A recent study by Pal et al. (2020) observed that the endophytic fungi Aspergillus aculeatus (strain C2) showed a maximum mycelial inhibition of 81.48%, and Crinipellis tabtim (strain M8) reduced disease incidence by up to 84.95% under field conditions in apple orchards. Furthermore, nonpathogenic R. necatrix isolates Rn12 and 29 have shown to successfully regulate R. necatrix pathogens (Arjona-López and López-Herrera, 2020). In another study, the antagonistic effect of T. harzianum against R. necatrix was inconsistent in greenhouses compared with laboratory evaluation results, and its efficacy was influenced by factors including root stock and soil type (Golafrouz et al., 2020). In pear orchards, disease occurrence appears to have increased in recent years, but reports on white root rot control in pear fruit trees using antagonistic fungi are absent. However, recent studies have suggested that T. harzianum could be a potential antagonist against R. necatrix.
Antagonistic Bacteria
Several studies have utilized bacteria and their metabolites in controlling white root rot (Table 1). These antagonistic bacterial species isolated from rhizosphere, soil, and plant endophytes were investigated as biocontrol agents against R. necatrix. Yasuda and Katoh (1989) observed that Agrobacterium and Pseudomonas, isolated from the soil or roots of peach and apple, colonize and survive efficiently, and play an important role in white root rot disease control. Similarly, bacteria isolated from the soil and rhizosphere of avocado orchards displayed a bicontrol effect against R. necatrix (Cazorla-Lopez et al., 2001; González-Sánchez et al., 2004). Yoshida et al. (2001) used bacterial metabolites derived from Bacillus amyloliquefaciens against R. necatrix, which strongly inhibited its growth in vitro. Kim et al. (2019) demonstrated that Bacillus thuringiensis C25 exhibited strong antagonistic effects on mycelial growth by degrading the cell wall structures of R. necatrix mycelia. Recently, Tienda et al. (2020) used a microbiome study to achieve an improved understanding of natural interactions between biocontrol agents and their impact on microbial communities. They observed that Pseudomonas chlororaphis (PCL1606) exhibited the highest biocontrol efficacy, and the biocontrol activity was directly correlated with the antifungal antibiotic 2-hexyl 5-propyl resorcinol, and the modified microbial communities. Despite several reports detailing the effective biocontrol agents against R. necatrix, biocontrol strategies have not been currently applied to control R. necatrix in pear orchards. Most of the biocontrol agents have been tested in other plant systems, and most studies have focused on in vitro or confined to greenhouse experiments, and their potential under field conditions needs further verification.
Virocontrol Using Mycoviruses
Biological control utilizing viruses is called virocontrol. Mycoviruses are virus-like organisms of double-stranded RNA (dsRNA) that infect fungi and reduce the pathogenicity of host fungi to their host plants. Mycoviruses have been the subject of research interest because of their potential as BCA against plant pathogenic fungi. Mycoviruses have also been proposed for the control of R. necatrix (Matsumoto, 1998; Matsumoto et al., 2002). Since R. necatrix spreads almost clonally and exclusively through root contacts, the pathogenic R. necatrix can be possibly transfected by placing the inoculum containing an effective dsRNA in contact with the mycelium of the pathogen, which then transfects the pathogen and spreads within the mycelial network. Considering the presence and distribution of virus-infected fungi, approximately 20% of those collected were found to be dsRNA-positive R. necatrix, and presumed to be infected by mycoviruses (Arakawa et al., 2002). Several studies on Japanese, Australian, Korean, Spanish, and Israelite isolates of R. necatrix describe the presence of dsRNA (Arakawa et al., 2002; Arjona-López et al., 2020; Chun and Kim, 2021). Importantly, several viruses such as Mycoreovirus 3 (MyRV3), Rosellinia necatrix megabirnavirus 1 (RnMBV1), and Rosellinia necatrix megabirnavirus 2 (RnMBV2), have been shown to induce hypovirulence in the R. necatrix fungal host (Chiba et al., 2009; Kanematsu et al., 2004; Sasaki et al., 2016). Recently, a member of the new viral species Rosellinia necatrix hypovirus 2 (RnHV2) infecting the pathogens showed lower colony growth in vitro and lower virulence in avocado plants than in virus-free R. necatrix (Arjona-López et al., 2021). Similarly, co-infection of R. necatrix by Rosellinia necatrix partitivirus 10 (RnPV10) and Rosellinia necatrix virga-like virus (RnVLV) confirmed the hypovirulence of R. necatrix (Arjona-López et al., 2020). By utilizing the successful example of virocontrol against R. necatrix, biological control of white root rot in pear orchards is achievable. For this purpose, R. necatrix must be isolated from the target orchard, interacted with mycoviruses containing a hypovirulence factor, and placed into the soil. The new virus-carrying strain can transfer the mycovirus as a donor to pathogenic strains in an orchard (Kondo et al., 2013).
Ecofriendly Methods for Disease Management
Ecofriendly disease management methods without the use of conventional chemical fungicides or bactericides are beneficial in managing plant diseases. Orchard growers have several management options that offer effective control of the pathogen with minimal impact on the environment. This can be achieved by interrupting the disease cycle, plant susceptibility, or the microbial balance, causing disease reduction below the economic injury level, rather than absolute control and maintenance of crop yield. Several “ecofriendly” plant disease management methods such as soil solarization, hot water treatment, weed management, and ecological engineering are available. These methods may not achieve 100% efficiency; however, in the orchards, they will attain a level of disease reduction that is below the economic injury level.
Solarization
Solarization is a hydrothermal process in which wet soil is covered with a transparent plastic sheet to absorb solar radiation (Pérez‐Jiménez, 2006). When the soil covered with a plastic sheet absorbs the radiation, the temperature of soil can increase up to 40–50°C, which affects soil pathogen due to exposure at high temperatures. This method developed in Israel during the late 1970s, was used as an ecofriendly approach to control white root rot disease. This method has proven effective and was used successfully against R. necatrix in established avocado and apple tree orchards (Freeman et al., 1990; López-Herrera et al., 1995; Sztejnberg et al., 1987) and apple nurseries (Sharma and Sharma, 2002). Sztejnberg et al. (1987) reportedly achieved 75% mortality of R. necatrix at a soil depth of 60 cm after 56 days of solarization. Furthermore, López-Herrera et al. (1999) reported complete destruction of the inoculum up to a depth of 60 cm after 6 weeks of solarization. Along with the thermal inactivation of the pathogen, other mechanisms, such as the accumulation of volatile substances under the plastic film and the activation of soil microorganisms assist in pathogen control (Freeman et al., 1990). Moreover, when analyzing solarized soils, Sharma and Sharma (2002) observed an increased population of thermo-tolerant antagonistic microorganisms, although the total microbial population, including fungi, bacteria, and actinomycetes decreased. However, soil solarization fulfills the requirements for successfully controlling soil-borne pathogen in orchards, as the trees are not damaged, since the inoculum is controlled at a considerable depth with delayed soil reinfestation.
Hot Water Treatment
Hot water treatment is an environmentally friendly method to control soil-borne diseases and offers fruit growers an alternative to chemical pesticides. Hot water treatment can be used in established orchards, to disinfect plant material used for propagation and kill the pathogen within the rhizosphere, exploiting the fact that R. necatrix is extremely heatsensitive. Hot water treatment was successfully used as a control strategy to prevent the spread of R. necatrix through infected tiger nut (Cyperus esculentus) tubers (Garcıa-Jiménez et al., 2004). The pathogen was completely devitalized in tubers treated at 55°C for 10, 20, or 30 min, and tubers did not show any reduction in sprouting. In field trials, hot-water treatments at 53°C or 55°C for 25 min provided good control of the disease, and normal plant development and yield were unaffected. Furthermore, Eguchi et al. (2008) dropped hot water on the rhizosphere of Japanese pear trees infested with the white root rot fungus R. necatrix, which eradicated the fungus from the colonized substrate when water at 35°C was provided for 3 days. The time required to eradicate R. necatrix decreased exponentially with increasing temperature and hot water treatment; white root rot mycelia on diseased roots were completely destroyed at 50°C and many rootlets grew after the treatment. Recently, Takahashi and Nakamura (2020) revealed the involvement of synergism between hot water and antagonism, with indigenous soil fungi such as T. harzianum in controlling R. necatrix. Furthermore, they selected commercialized Trichoderma products that enhanced soil antagonism and determined how to use them in combination with hot water to eradicate white root rot under field conditions (Takahashi et al., 2020). However, in conclusion, this environmentally safe method is applicable to orchards of fruit trees that can tolerate a minimum water temperature of 45°C.
Weeds Management in Orchards to Control Disease
Weeds are common on the floors of pears or fruit orchards, and their roots remain alive for a certain time period even after trimming. They may serve as reservoirs for pathogens and their vectors as alternative hosts (Wisler and Norris, 2005). Sztejnberg and Madar (1980) reported that R. necatrix attacked and killed several weed species, such as Prosopis farcta, Amaranthus gracilis, and Conyza bonariensis, suggesting that weeds can promote the spread of the disease. Furthermore, Shiragane et al. (2019) observed that nine out of the 20 weed species collected from two pears (Pyrus pyrifolia) orchards were infested with the fungus R. necatrix, without exhibiting any disease symptoms. They reported that rescue grass (Bromus catharticus) facilitates the spread of R. necatrix, and some weeds may be able to supply nutrients for the mycelial growth of this pathogen in their rhizosphere. These studies suggest that weeds play an important role in the propagation of the disease in orchards. However, certain weed species can possibly inhibit or act as antagonists to the growth and spread of R. necatrix. An unpublished study by Lee (2002) observed that Poa pratensis grass had a strong inhibitory effect on the incidence of white root rot disease by sustaining the growth of antagonistic microflora. Therefore, there is an urgent need to study potential host weeds, either antagonists or spreaders, which can be strategically applied as a biocontrol agent against R. necatrix.
Ecological Engineering for White Root Rot Disease Management
Ecological engineering has recently emerged as an ideal example of a pest management approach that relies on the application of cultural practices that affect habitat manipulation, enhancing biological control (Nayak et al., 2018). Habitat manipulation can be achieved by creating a suitable ecological environment within the agricultural field for natural enemies by providing food resources, alternative hosts/prey, and shelter under adverse conditions. The main focus of ecological engineering is to reduce the mortality rate of natural enemies by providing supplementary resources and manipulating host plant traits for the benefit of natural biocontrol agents. There is a growing realization that soil-borne diseases can be managed with exploitation and interventions of microbes and their metabolites, in addition to selecting appropriate plant varieties. However, ecological engineering for below-ground disease management, such as white root rot caused by fungus, can be achieved by the following activities to increase the beneficial microbial population and enhance soil fertility in orchards.
1) Practices that reduce humidity beneath the canopy, through adequate pruning regimes and an adequate drainage system, treating soil with lime to increase soil pH, and the removal of coarse woody debris, will help to control the disease and reduce the risk of infection.
2) Remove the remains of dead trees, especially their roots, and organic material that has been infected with R. necatrix, and burn them on-site.
3) Good soil conditions, effective drainage, balanced fertilization, and balanced growth are of major importance in preventing root infections caused by soil-borne fungal diseases.
4) Add organic matter in the form of farm yard manure, vermicompost, and crop residue, which enhances the below-ground biodiversity of beneficial microbes.
5) Application of balanced dose of nutrients using biofertilizers based on soil test report.
6) Application of biofertilizers with a special focus on mycorrhiza and plant growth promoting rhizobia.
7) Application of Trichoderma harzianum/viride and Pseudomonas fluorescens for the treatment of seed/seedling/planting materials in nurseries, and subsequent field applications.
Future Prospects
The potential of bacteria, fungi, and mycoviruses in controlling soil-borne plant diseases is enormous, yet better strategies should be developed to holistically exploit their benefits. The application of biocontrol agents at the field or orchard level is not well-characterized, and reports on their effects at the field level is limited. Currently, most studies focus on the characterization and testing of biocontrol agents in vitro or in confined greenhouse experiments, and their potential under field conditions requires further verification. Much effort should be devoted to further characterize and implement it in the field. A multi-strain consortia study of biocontrol agents, which is closer to natural conditions, than the mass application of a single microbial strain will benefit in the future. Microbiome analysis and characterization of endophytic bacteria to identify disease resistance-conferring agents will certainly play an important role in further improving biological control. Ecological engineering using multi-strain consortia can provide a new paradigm for soil-borne disease control, which can facilitate the establishment of more sustainable agricultural applications.
Notes
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Acknowledgements
This work was supported by a grant from the National Institute of Horticultural and Herbal Science (NIHHS) funded by the Rural Development Administration (RDA) of the Republic of Korea (PJ01448401).