Antibiotic Resistance of Pectobacterium Korean Strains Susceptible to the Bacteriophage phiPccP-1
Article information
Abstract
Commercial products with antibiotics like streptomycin as active ingredients have been used to control soft rot disease caused by Pectobacterium species for a long time. In this study, antibiotic resistance of twenty-seven Korean strains of Pectobacterium species including P. carotovorum, P. odoriferum, P. brasiliense, and P. parmenteri, which were previously shown to be susceptible to the bacteriophage phiPccP-1 was surveyed using a disk diffusion assay. While all strains were highly susceptible to ampicillin, kanamycin, chloramphenicol, tetracycline, and rifampicin, some strains showed weak susceptibility to 300 μg/ml of streptomycin. Furthermore, some of them are partially or completely resistant to commercial pesticides—Buramycin and streptomycin at the concentration of 250 μg/ml that is recommended by the manufacturer for streptomycin-based pesticides. These results indicate the presence of streptomycin-resistant Pectobacterium strains in South Korea, and the development of antibiotic alternatives to control soft rot is needed.
Plant-pathogenic bacteria belonging to the Pectobacterium genus (formerly Erwinia, Winslow et al., 1920) cause soft rot disease in diverse plants. They are considered as broad-host-range pathogens because they have been isolated from so many plant species, and individual strains are pathogenic to various plant species under experimental conditions (Ma et al., 2007). Furthermore, they have also been found in association with a variety of invertebrates (Glasner et al., 2008). Several different plant cell wall degrading enzymes are produced by Pectobacterium as major virulence factors which results in soft rot symptom in host plants. These bacterial pathogens cause severe economic losses on agricultural, horticultural, and ornamental plants during vegetation, transportation, and storage.
The genus Pectobacterium currently carries 18 species (Jee et al., 2020; Lee et al., 2021). Among them, P. odoriferum, P. carotovorum, P. brasiliense, and P. versatile are dominant species found in potato and napa cabbage fields in South Korea (Jee et al., 2020). In addition, global warming might result in the population changes of dominant species in South Korea. These pathogens are spread by various ways, including wa-ter, seeds, equipment, and insect vectors. Therefore, avoiding contamination, using healthy plant material, and rotating crops have been applied as control strategies against soft rot. Additionally, physical and chemical treatments have been widely used (Czajkowski et al., 2011).
Antibiotics have been used to control various bacterial diseases in plants since the late 1950s. Currently, streptomycin and oxytetracycline are most commonly used in the control of plant diseases (McManus et al., 2002). Additionally, streptomycin was considered a promising control agent for black leg and soft rot diseases for a long time (Czajkowski et al., 2011; Yongsheng et al., 2014). However, the overuse of antibiotics results in the emergence of antibiotic-resistant bacteria. In the case of streptomycin, streptomycin-resistant plant and ani-mal pathogens were reported. For example, 24 of 367 isolates of Mycobacterium tuberculosis from the United States in 1994 were resistant to 500 µg/ml of streptomycin (Cooksey et al., 1996). The streptomycin resistance was also found in plant pathogens such as Erwinia amylovora (Coyier and Covey, 1975; Miller and Schroth, 1972), Pseudomonas syringae pv. papulans (Burr et al., 1988; Jones et al., 1991), P. syringae pv. syringae (Sundin and Bender, 1993; Young, 1977), Xanthomonas campestris pv. vesicatoria (Minsavage et al., 1990), etc. Currently, streptomycin resistance in plant pathogens has been reported more and more (Han et al., 2003; Lee et al., 2020; Russo et al., 2008; Xu et al., 2010). Natural streptomycin resistance in some Pectobacterium species, previously named as Erwinia carotovora subspecies, causing diseases in tobacco and Japanese radish, has also been reported (Kang et al., 1989; Kobayashi et al., 1987). However, how widely streptomycin resistance is present in Pectobacterium species isolated in South Korea has not been well studied.
In this study, the antibiotic resistance or sensitivity of various Pectobacterium strains isolated in South Korea was evaluated against 6 common antibiotics, including streptomycin, using a disk diffusion assay (Table 1, Fig. 1). Twenty-seven strains, 14 strains of P. carotovorum, 5 strains of P. brasiliense, 7 strains of P. odoriferum, and 1 strain of P. parmenteri previously shown to be susceptible to the bacteriophage phiPccP-1 (Lee et al., 2021), were evaluated in this study. Six antibiotics, ampicillin, chloramphenicol, kanamycin, tetracycline, rifamycin, and streptomycin, were selected for this study and treated on paper disks at their common concentration: 1 mg/ml, 3 mg/ml, 3 mg/ml, 3 mg/ ml, 500 μg/ml, and 300 μg/ml, respectively. For a disk diffusion assay, 10 μl of antibiotics except streptomycin was dropped on filter paper disks placed on Luria-Bertani (LB) plates inoculated with 107 cfu/ml of bacterial suspension of Pectobacterium bacteria, and then plates were incubated at 26° C overnight. To avoid the eclipse of paper disks, 10 μl of the streptomycin was dropped directly on plates. The results showed that all tested strains showed high susceptibility to ampicillin, streptomycin, chloramphenicol, kanamycin, tetracycline, and rifamycin (Table 1, Fig. 1). Notably, the streptomycin susceptibility was different among those strains. Some strains showed pale and weak susceptibility against streptomycin (Fig. 1).
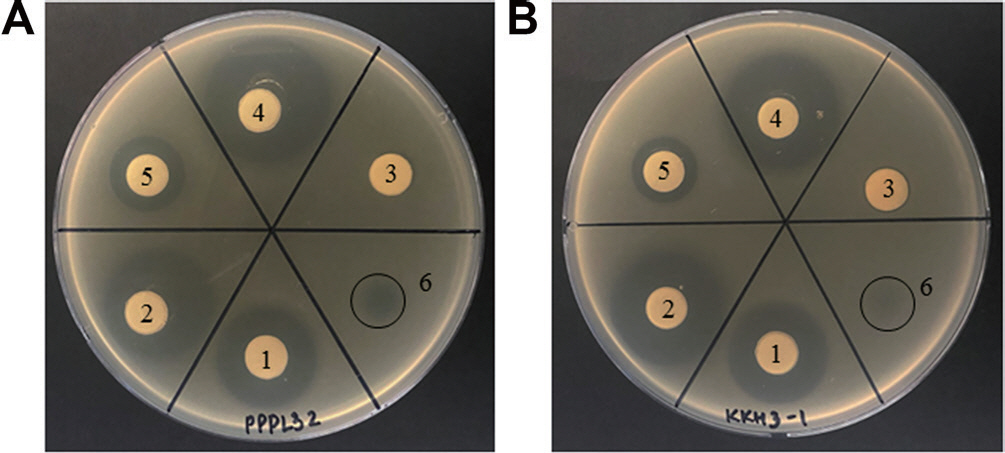
Resistance or susceptibility of representative Pectobacterium strains against 6 antibiotics. After bacterial growth (A, PPPL32 strain; B, PcKKH3-1 strain) on Luria Bertani plates, 10 μl of the fol-lowing concentration of each antibiotic was dropped on paper disks. Black circles highlight dropped sites of streptomycin. 1, ampicillin (1 mg/ml); 2, chloramphenicol (3 mg/ml); 3, kanamycin (3 mg/ml); 4, tetracycline (3 mg/ml); 5, rifamycin (500 μg/ml); 6, streptomycin (300 μg/ml).
The minimum inhibitory concentration (6.25 μg/ml) of streptomycin sulfate against P. carotovorum was previously reported (Nguyen et al., 2017). P. carotovorum also showed the susceptibility to filter paper disks dipped in 200 ppm of streptomycin (Gokul et al., 2019). Foliar application of streptomycin products like Agreptor (GyungNong, Seoul, Korea), Buramycin (Farm Hannong, Seoul, Korea), and Agrimycin (SUNGBO Chemicals, Seoul, Korea) has been used to control the soft rot disease caused by P. carotovorum (Bhat et al., 2017; Sang et al., 2015) and P. odoriferum (Lee et al., 2021). The manufacturer-recommended concentration of representative streptomycin products to control the soft rot disease in Korea are 250 μg/ml for Agreptor and Buramycin and 75 μg/ml for Agrimycin (Abd-El-Khair and Karima, 2007; Shrestha et al., 2009). Therefore, we also check whether Pectobacterium strains can be resistant to streptomycin at the concentration of the commercial antibiotic, 250 μg/ml. Instead of using filter paper dishes, 10 μl of 250 μg/ml of streptomycin itself or a commercial streptomycin-based commercial pesticide, Buramycin were dotted directly into LB plates inoculated with bacterial suspension. As expected, most of the tested strains showed susceptibility to both streptomycin itself and Buramycin with different patterns (Table 2, Fig. 2). Six tested strains showed strong susceptibility to both streptomycin and Buramycin, while 16 strains showed mild susceptibility to either streptomycin itself or Buramycin. Among them, some strains are resistant to streptomycin itself, but mild susceptibility to Buramycin. Notably, 5 strains including 4 strains of P. carotovorum and 1 strain of P. brasiliense showed strong resistance to both streptomycin itself and Buramycin. These results indicate the emergence of streptomycin resistance in Pectobacterium populations in South Korea, which is not only limited in single species but also various species.
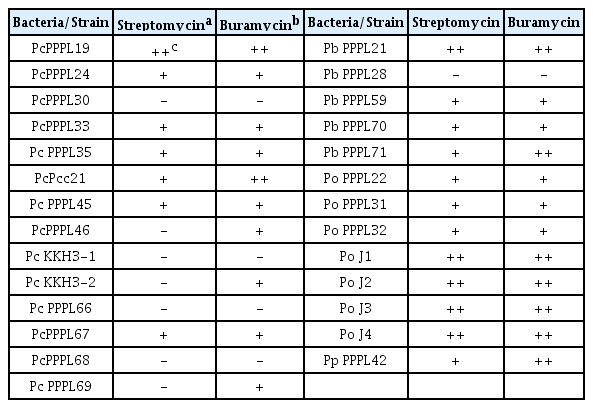
Resistance or susceptibility of Pectobacterium strains against Buramycin and streptomycin as the equal amount to the manufacturer-recommended concentration of streptomycin in Buramycin
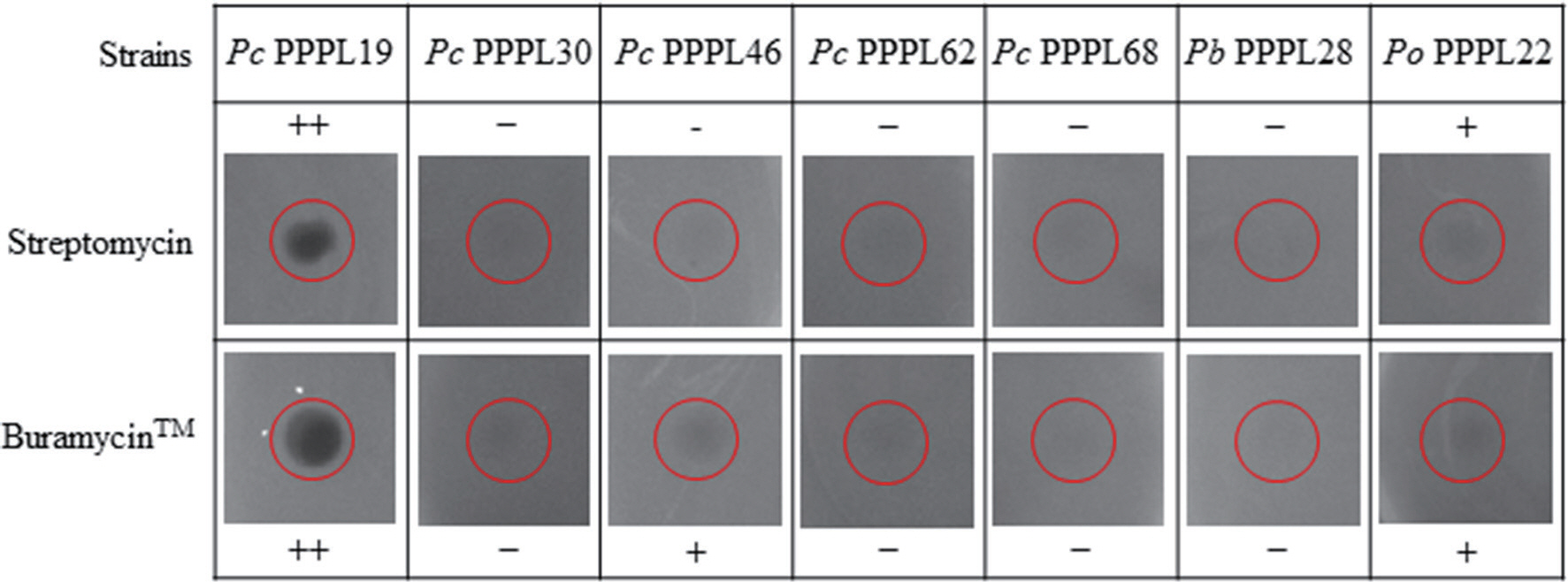
Resistance or susceptibility of several Pectobacterium strains against streptomycin itself and streptomycin-based product, Buramycin, at the concentration of 250 μg/ml. Red circles highlight dropped sites of streptomycin itself or Buramycin. Pc, P. carotovorum; Pb, P. brasiliense; Po, P. odoriferum. ++, highly susceptible; +, mild susceptible; -, resistant.
Additionally, the distribution of resistant strains seems not related to isolated host plants or location or even time of isolation. Indeed, some strains such as PPPL45‒46, PPPL62‒63, PPPL66‒69 showed different susceptible patterns against streptomycin itself and commercial antibiotic product, although they were isolated at same time, same location and same plant species. Furthermore, those strongly or partly resistant strains were isolated from various plants such as cabbage, potato, kiwi, and tomato, which are severely damaged by Pectobacterium species (Abd-El-Khair and Karima, 2007; Czajkowski et al., 2011; Dees et al., 2017; Lee et al., 2021). Notably, the streptomycin resistance of strains isolated in 1997 indicates that the streptomycin resistance already existed a long time ago. Therefore, natural existence of streptomycin resistance in Pectobacterium species should be considered for disease control practice in the future.
In conclusion, Pectobacterium strains isolated in South Korea are still susceptible to several antibiotics, while some of them are resistant to streptomycin. The antibiotics products carrying streptomycin as an active ingredi-ent have been used to control diverse bacterial diseases for a long time. This might result in the introduction of streptomycin resistance in Pectobacterium strains. This information will alert us to develop alternative methods for control of soft rot caused by Pectobacterium strains in the future such as biological control or other antibacterial compounds.
Notes
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Acknowledgments
This work was carried out with the support of the “Coop-erative Research Program for Agriculture Science & Technol-ogy Development (Project No. PJ014219022022)” of the Rural Development Administration, Republic of Korea.

