Screening of Bacterial Antagonists to Develop an Effective Cocktail against Erwinia amylovora
Article information
Abstract
Several types of chemical bactericides have been used to control fire blight. However, their excessive usage leads to environmental deterioration. Therefore, several researchers have analyzed antagonistic microorganisms as promising, effective, and safe biological control agents (BCAs). The primary aim of this study was to screen for potential antagonistic bacteria that suppress Erwinia amylovora. Among the 45 isolates studied, 5 strains showed the largest inhibition zone against E. amylovora. 16S rRNA gene sequencing identified them as Bacillus amyloliquefaciens (KPB 15), B. stratosphericus (KPB 21), B. altitudinis (KPB 25), B. safensis (KPB 31), and B. subtilis (KPB 39). KPB 25 and 31 reduced the lesion size of fire blight by 50% in immature apple fruits, and did not show antagonism against each other. Therefore, KPB 25 and 31 were selected to develop an antagonistic mixture against fire blight. Although the mixture with KPB 25 and 31 showed a slightly increased ability to reduce lesion size on immature fruits, they did not exhibit a synergistic effect in reducing E. amylovora population compared to each strain alone. Nevertheless, we have identified these two strains as useful and novel BCAs against fire blight with additional benefits safety and potential in developing a mixture without loss of their activity, owing to the absence of antagonism against each other.
Introduction
The occurrence of fire blight caused by Erwinia amylovora in apple and pear trees in Korea showed regularity in terms of newly infected orchards that were contaminated in the vicinity of already infected orchards. However, despite being a long distance from the infected orchards, fire blight made an unexpected appearance in many newly infected orchards. Therefore, the management of the spread of E. amylovora has been unsuccessful most likely due to its several modes of dissemination, including natural factors such as wind-driven rain (McManus and Jones, 1994) and honeybees (Choi et al., 2022a), and artificial factors such as the movement of workers and agricultural equipment (Choi et al., 2019; van der Zwet et al., 2012). Therefore, active management strategies must be developed, based on the current dispersal patterns in Korea, to prevent the spread of this pathogenic bacterium.
Commercial products including 12 antibiotics and 8 copper compounds, either as single antibiotics (streptomycin, oxytetracycline, oxolinic acid, and validamycin) and copper compounds (copper hydroxide, copper sulfate basic, tribasic copper sulfate, and copper oxychloride), or their mixtures have been registered by the Rural Development Administration of Korea (RDA) as active ingredients of commercial products for the fire blight control (Park et al., 2017). The use of antibiotics for plant protection has been either not permitted or prohibited in the European Union, United States, and Canada due to the increased risk of resistance problems and adverse environmental impacts (Fried et al., 2013; Mikiciński et al., 2020). However, Korea has permitted their use. The use of copper compounds is also limited because they only have a protective effect but not a curative effect, and also have a side effect of the formation of russet on the leaves and fruits of apples (Mikiciński et al., 2020). Thus, biological control agents (BCAs) developed using antagonistic bacteria effective against E. amylovora has garnered increasing attention as a potential resources of controlling the fire blight. To date, several BCAs, including the genera Pseudomonas: P. fluorescens A506 (Wilson and Lindow, 1993) and EPS62e (Cabrefiga et al., 2007), P. orientalis F9 (Zengerer et al., 2018), P. vancou-verensis L16, P. chlororaphis 3M, P. congelans 35M. P. protegens 59M (Mikiciński et al., 2020); the genera Pantoea: P. agglomerans E325 (Pusey, 2002) and P10c (Vanneste et al., 2002), P. vagans C9-1 (Ishimaru et al., 1988; Smits et al., 2010); the genera Bacillus: B. subtilis QST713 (Aldwinckle et al., 2002) and BD170 (Broggini et al., 2005). B. amyloliquefaciens LMR2 (Bahadou et al., 2018), MB40 (Shemshura et al., 2020), and FZB42 (Chen et al., 2009); Paenibacillus polymixa N179 (Fallahzadeh-Mamaghani et al., 2021); Lactobacillus plantarum (Roselló et al., 2013); Enterobacter sp. En113 and Serratia sp. Se111 (Sharifazizi et al., 2017); and the fungal BCA Aureobasidium pullulans (Kunz, 2006), have been reported to have potential use against fire blight worldwide. Among these, several BCAs have been commercialized as biopesticides, such as BlightBan A506 (P. fluorescens A506, USA), Bloomtime (P. agglomerans E325, USA), BlossomBless (P. agglomerans P10c, New Zealand), BlightBan C9-1 (P. vagans C9-1, USA), Serenade (B. subtilis QST713, USA), Biopro (B. subtilis BD170, Switzerland), and BlossomProtect (A. pulllans, Germany). In Korea, two biopesticides, Serenade (B. subtilis QST713, USA) and Serifel (B. amyloliquefaciens MBI600, USA) (Fravel, 2005), have recently been registered by the RDA for fire blight control.
However, the efficacy of biopesticides for fire blight control is controversial because their effects vary depending on the environmental conditions and disease severities in each orchard (Reininger et al., 2019). Therefore, in order to succeed in fire blight control using BCAs, it is necessary to intensive research on mechanisms of action and targets of BCAs will be required in Korea beside other countries. Biopesticides for fire blight control may be permitted in Korea if the novelty and biosafety of their microbial sources are proven. Recently, few studies have been conducted to develop domestic BCAs using antagonistic bacteria (Kim et al., 2022), lichen extracts (Lee et al., 2021), and bacteriophages (Park et al., 2018, 2022). Although these BCAs were developed domestically to en-sure originality, they came from a single source. To satisfy originality, in this study, we isolated antagonistic bacteria from different apple tissues, collected in Korea to satisfy originality, and tested their effectiveness in suppressing fire blight. Furthermore, cocktails of various antagonistic bacteria were investigated in order to maximize the effect of controlling fire blight.
Materials and Methods
Isolation of antagonistic bacteria against E. amylovora.
The antagonistic bacteria used in this study were isolated from blossoms, leaves, and shoots of apple trees (Malus domestica Borkh. cv. Fuji), and soil samples collected from apple orchards in Chuncheon, Hoengseong, Hwacheon, Inje, and Sangju, Korea, in 2021. Two flowers and 2 cm sections of the uppermost portion of shoots and petiole of leaves were sep-arately sterilized twice with 70% ethanol for 5 min, washed in sterilized water, and then homogenized with 2 ml of 10 mM MgCl2 buffer in an extraction bag (Bioreba, Reinach, Switzerland). Soil (1 g) was mixed with 5 ml of 10 mM MgCl2 buffer in a 15 ml tube without any sterilization. Each 1 ml aliquot of extract from flower, leaf, shoot, and soil was serially diluted, and 100 µl of the 10−2 dilution was plated on Luria-Bertani (LB) medium and incubated at 28°C for 3 days.
Inhibition zone assays were performed to screen for antagonistic bacteria against E. amylovora (Shemshura et al., 2020). Culture plates with the media were pre-plated with E. amylovora TS 3128 strain (Kang et al., 2021) 12 hr before spot inoculation of the isolated colonies, followed by incubation at 28°C for 3 days. Isolated bacterial colonies of different sizes, colors, and morphologies were spotted on the media and showed the inhibition zones produced by the isolated colonies. The media used for inhibition zone assays were as follows: LB (tryptone 10 g, yeast extract 5 g, NaCl 10 g, 1 M NaOH 1 ml, agar 18 g/l), King's B (KB: protease peptone 20 g, MgSO4·H2 O 0.4 g, glycerol 10 ml, K2 HPO4 1.5 g, agar 18 g/ l), mannitol glutamate yeast extract (MGY: mannitol 10 g, L-glutamic acid 2 g, KH2 PO4 0.5 g, NaCl 0.2 g, MgSO4·7H2 O 0.2 g, agar 18 g/l), tryptic soy agar (pancreatic digest of casein 15 g, papaic digest soybean meal 5 g, NaCl 5 g, agar 15 g/l), R2A (yeast extract 0.5 g, peptone 0.5 g, casein hydrolysate 0.5 g, starch 0.05%, glucose 0.5 g, K2 HPO4 0.3 g, MgSO4·H2 O 0.3 g, NaCl 0.3 g, agar 15 g/l), and N-acetylglucosamin acid agar (nutrient broth 23 g, glycerol 16 ml, agar 18 g/l). Antagonism was determined as the diameter of the zone where the growth of E. amylovora was inhibited. Finally, the representative antagonistic bacteria that showed relatively larger diameter of inhibition zones and inhibitory effect on one or more media were selected.
To evaluate the activity of the isolates did not form inhibition zones on medium such as KB, TS 3128 harboring the kanamycin resistance gene was first plated on KB media, followed by spot inoculation of the isolates. After incubation at 28°C for 2 days, a section 1.5 cm away from the overgrowth region by motility from center of inoculation was collected using no. 1 cork borer (5 mm), macerated in 1 ml of 10 mM MgCl2 buffer, and then plated on KB medium supplemented kanamycin (50 µg/ml). After incubation at 28°C for 1 day, the number of colonies was counted as colony forming units (cfu/ml). Inhibition assay was then performed twice on the cfus in the overgrowth region.
Identification of antagonistic bacteria.
Partial 16S rRNA gene sequences were used to determine the phylogeny of five representative antagonistic bacteria selected in this study. The antagonistic bacteria were grown in LB broth at 28°C for 2 days, bacterial pellets were collected after centrifu-gation at 3,000 ×g for 10 min, and genomic DNA was extract-ed using the Wizard Genomic DNA purification Kit (Promega, Madison, WI, USA), following the manufacturer's instructions. Polymerase chain reaction (PCR) was performed using the primers fD1 and rP2 to obtain an approximately 1.5 kb am-plicon (Weisburg et al., 1991). The amplicons were eluted using 1% agarose gel electrophoresis and were cloned into the TOPO TA cloning vector (Invitrogen, Carlsbad, CA, USA), and sequenced. Each sequence was compared using BLAST to identify the species and MEGA XI with the neighbor-joining method with 1,000 bootstrap replicates was used to con-struct the phylogenetic trees (Shemshura et al., 2020).
Analysis of antagonism in the antagonistic bacteria.
To analyze antagonism among the five representative antagonistic bacteria, each strain was spotted on the MGY medium that was pre-plated with the other four antagonistic bacteria before 12 hr, incubated at 28°C for 3 days to observe the inhibition zone produced by the spotted strain. Antagonism among the antagonistic bacteria was determined by the production of an inhibited zone of growth of the pre-plated strain, which was also regarded as scarring around the strain inoculated by spotted antagonistic bacteria.
Efficacy of antagonistic bacteria against fire blight in immature apple fruits and seedlings.
The biological control efficacies of the five representative antagonistic bacteria were evaluated in immature apple fruits and seedlings. Half-sectioned immature apple fruits (cv. Fuji) were sterilized three times with 2% sodium hypochlorite and cyclohexi-mide (2 µg/ml) for 8 min, respectively, washed in distilled water, and punctured using a no. 1 cork borer. These half-sectioned fruits were then dipped into a single suspension (O.D.600nm=0.1, approximately 105 cfu/ml) or a mixture of two antagonistic bacteria with the same concentration supplemented with 0.02% Silwet L-77 (Lehle Seeds, Round Roc, TX, USA) for 20 min and dried on a filter paper. For inoculation of E. amylovora TS 3128 harboring a kanamycin resistance gene in the pBAV1K vector, a 20 µl bacterial suspension (O.D.600nm=0.1) was inoculated into the hole, followed by incubation at 28°C for 6 days. Subsequently, the lesion size was measured to evaluate the biocontrol efficacy of antagonistic bacteria. In addition, a 1 cm2 portion that was collected using a no. 4 cork borer including the inoculated hole at day 6, and macerated in 10 mM MgCl2 buffer, and 100 µl of its suspension was plated on MGY medium supplemented with kanamycin (50 µg/ml) and incubated at 28°C for 2 days. The number of colonies of E. amylovora TS 3128 that grew on the plates were counted as cfu/ml. In addition, the efficacy of the mixtures of two selected antagonistic bacteria was tested based on the reduction in lesion size and E. amylovora TS 3128 population on immature apple fruits. Each observation was collected using six half-sectioned fruits with two replicates. E. amylovora TS 3128 colonies were con-firmed via colony-PCR using specific primers (Powney et al., 2011).
Four-week-old apple seedlings (M. domestica cv. M9) were dipped in the antagonistic bacterial suspension under the same conditions as the fruits, subsequently inoculated with E. amylovora TS 3128 harboring pBAV1K by spraying three times, and incubated at 28°C for 6 days. The efficacy of the antagonistic bacteria against fire blight on apple seedlings was analyzed according to the disease severity rank from 0 to 5: 0, no symptoms; 1, only oozing out of lesion; 2, browning of upper leaves and oozing; 3, start of necrosis on upper leaves; 4, necrosis and browning on lower leaves; and 5, necrosis in half of the seedlings. Each observation was collected from five seedlings, with two replicates each.
Statistical analysis.
Two replicates of the experiments were performed to determine antagonism using inhibition zone test and in planta assays. Data were analyzed using analysis of variance (ANOVA), and means were compared using Duncan's least significant difference test at significance levels of P<0.05. In addition, box and whisker plot was used to show histogram analysis provided a sufficient display in reduction of lesion size on immature apple fruits. The analysis was performed using the SAS software (version 9.2, SAS Institute Inc., Cary, NC, USA).
Results
Screening of antagonistic bacteria against E. amylovora.
In total, 45 colonies with different sizes, colors, and morphologies were isolated and viz. the Kangwon Plant Pathogenic Bacteria (KPB) strains (Table 1). Among the 45 strains, 12 (KPB 07, 11, 13, 15, 19, 21, 25, 27, 31, 34, 39, and 41) showed inhibition zones (1 mm or more in diameter) against E. amylovora TS 3128 on MGY medium, whereas the other strains did not show any inhibitory effects against E. amylovora TS 3128, regardless of medium types (Table 1). Finally, five strains (KPB 15, 21, 25, 31, and 39) were chosen as representative based on the relatively larger diameter of their inhibition zones compared to that of the other strains, and their inhibitory ability on medium types besides MGY medium (Table 1, Fig. 1A).

Isolation sources and locations of BCAs and their inhibitory activity against Erwinia amylovora TS 3128 on different media

Antagonistic activity of the five representative strains (KPB 15, 21, 25, 31, and 39) against Erwinia amylovora TS 3128. (A) Diameter of inhibition zone against TS 3128. (B) Cfu of TS 3128 under the overgrowth of these strains on King's B (KB) medium. Control indicated cfu of TS 3128 plated alone on KB medium. Results presented are those combined from two replicates. Data with different alphabets indicate significantly different means (P<0.05).
In addition, the growth of the strains KPB 15 and 39 reached 9 and 6 cm from the center of inoculation on KB medium, respectively, indicating that these strains have higher motility than other strains. The inhibition zone could not be measured due to the masking of TS 3128 growth by the overgrowth of KPB 15 and 39. However, TS 3128 population was significantly lower in the overgrowth area of the KPB 15 and 39 strains than that in the overgrowth area of KPB 21, 25, and 31 (Fig. 1B).
Phylogenetic analysis of antagonistic bacteria.
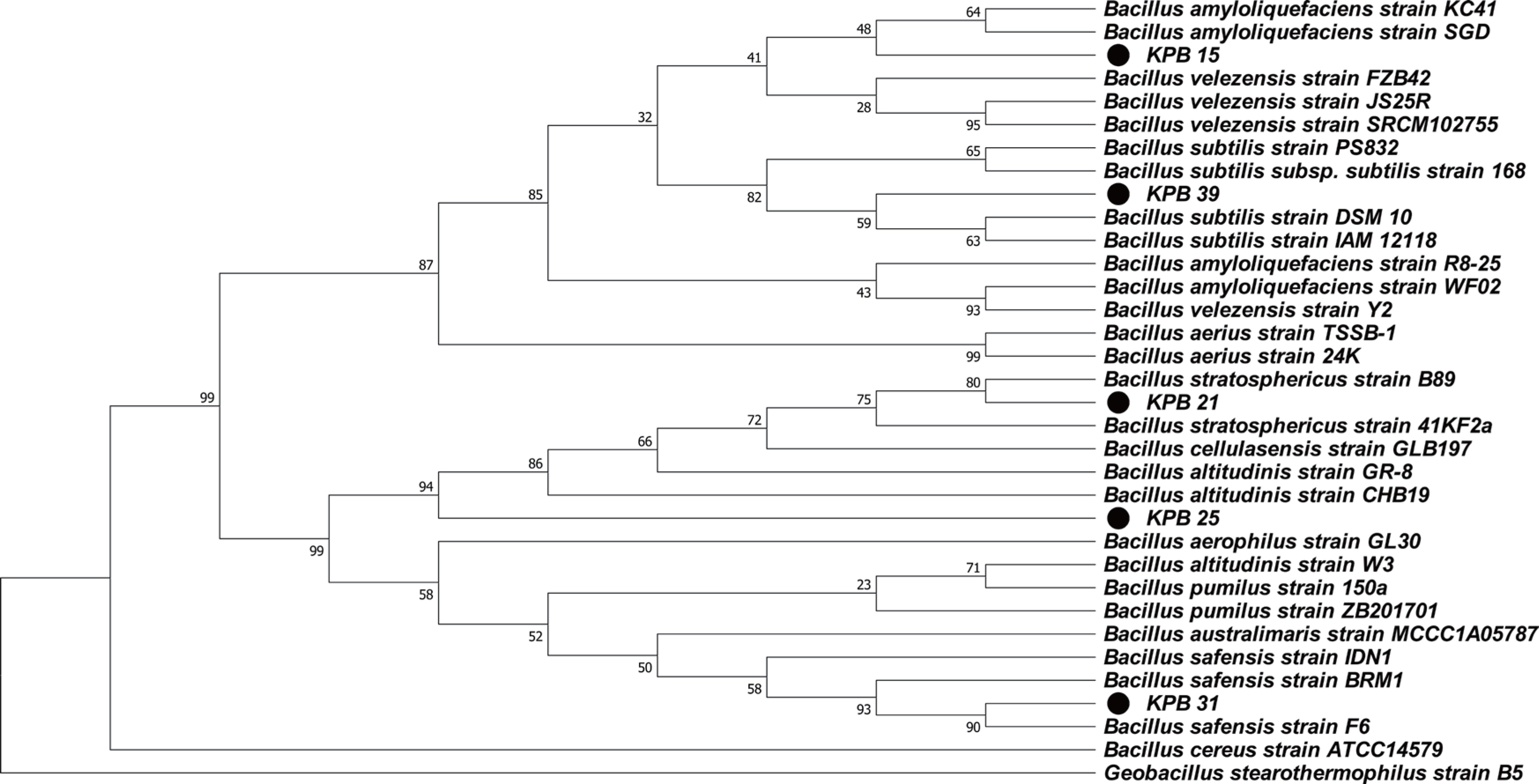
To determine the phylogenetic relatedness of the five representative antagonistic bacteria against E. amylovora, a phylogenetic tree was constructed using the 16S rRNA gene sequences of total of 29 valuable Bacillus species, including 12 antagonistic species, two human and plant pathogenic species, 13 species unrelated to antagonism and pathogens, and 1 Geobacillus species as an out-group. The species closest to the five antagonistic bacteria (KPB 15, 21, 25, 31, and 39) were B. amyloliquefaciens KC41, B. stratosphricus 41KF2a, B. altitudinis CHB19, B. safensis BRM1, and B. subtilis DSM 10, with a similar-ity of 99.6%, 99.9%. 99.7%, 99.8%, and 99.5%, respectively (Fig. 2). None of the five antagonistic bacteria clustered with either of the two animal or plant pathogens.
Antagonism test among the antagonistic bacteria.
KPB 15, 21, 25, and 31 did not show any antagonism against the other 4 KPB strains. However, KPB 39 produced an inhibition zone around the spot inoculation point on pre-plated with KPB 21, 25, and 31, indicating that KPB 39 inhibited KPB 21, 25, and 31 growth (Table 2).
The fire blight control efficacy of antagonistic bacteria in immature apple fruits and seedlings.
The biological control efficacy of the five selected antagonistic bacteria in this study was shown to be greater in seedlings than that in immature fruits. In immature apple fruits, KPB 25 and 31 significantly reduced the lesion size (approximately 0.7 cm reduction), and KPB 39 showed moderate antagonism against E. amylovora TS 3128 (Fig. 3A). In contrast, KPB 15 and 21 did not reduce the lesion sizes compared to the negative control, E. amylovora TS 3128 alone. However, all five KPB reduced the disease severity by 20% in apple seedlings (Fig. 3B). Therefore, based on the lack of reduction of lesion size on immature fruits by KPB 15 and 25, and antagonism of KPB 39 against other strains (KPB 21, 25, and 31 strains), we chose the strains KPB 25 and 31 to develop a cocktail as a BCA against fire blight.

In planta assays of the five representative strains (KPB 15, 21, 25, 31, and 39) for suppression of fire blight. (A) Necrotic lesion size on immature apple fruits (Fuji) when the 5 biological control agents (BCAs) were inoculated 1 hr prior to treatment with TS 3128. (B) Disease severity based on index using oozing, browning of leaves, and necrosis on apple seedlings (M9) following the application of the five BCAs prior to TS 3128 inoculation. Results presented are those combined from two replicates. Data with different alphabets indicate significantly different means (P<0.05).
In double mixtures of KPB 25 and 31, or KPB 25 or 31 each with KPB 15 or 21, we observed no significant synergistic effects on the reduction of lesion size on immature apple fruits when compared to that by KPB 25 or 31 alone, as evidenced by the sum and standard deviation of lesion size. However, based on the analysis using box and whisker plot method, the mixture of KPB 25 and 31 showed significantly reduced lesion size compared to that in the single antagonistic bacterial treatments (Fig. 4A). However, in population test, no synergistic effects were observed in any of the double mixtures (Fig. 4B)
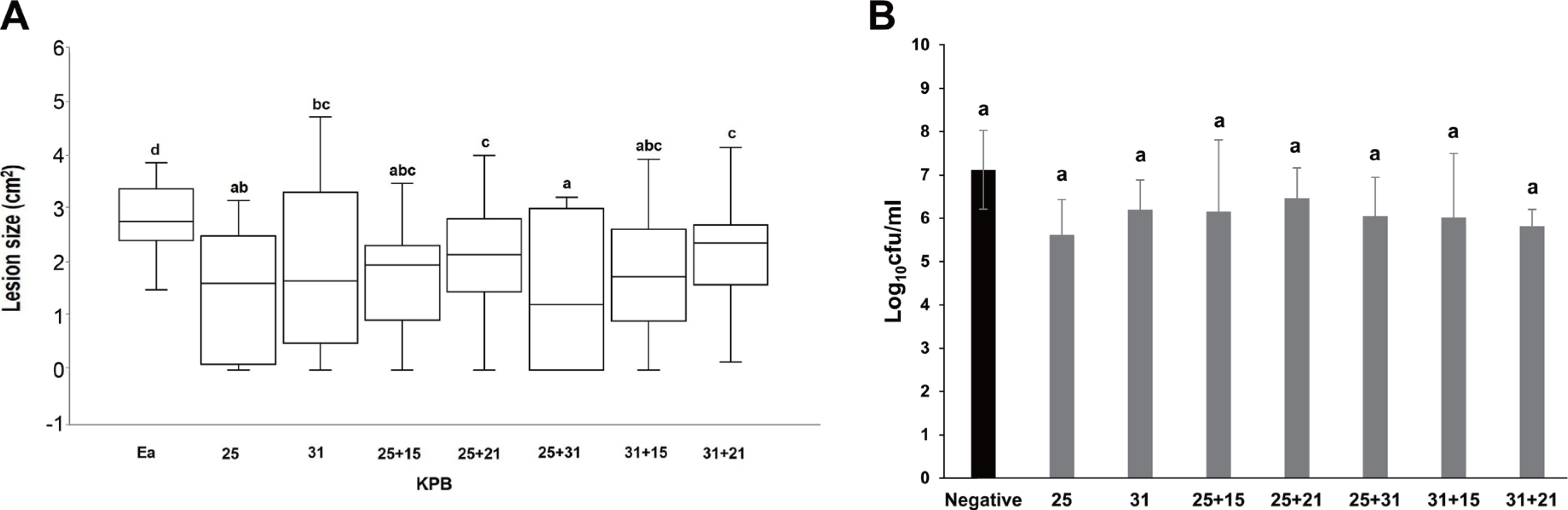
In planta assays of double mixtures of KPB 25 and 31, or each KPB 25 or 31 strain with KPB 15 or 21 strain for the suppression of fire blight. (A) Necrotic lesion size on immature apple fruits (Fuji) when double mixtures were inoculated 1 hr prior to treatment with TS 3128. (B) Population of TS 3128 on immature apple fruits inoculated with double mixtures were 1 hr prior to treatment with TS 3128. Results presented are those combined from two replicates. Data with different alphabets indicate significantly different means (P<0.05).
Discussion
In Korea, fire blight was first observed in 2015, has since remained a devastating disease in apple and pear orchards because the infected trees or orchards with >5% disease severity must be buried to reduce inoculum levels and prevent further spread of the disease. Moreover, it is not easy to erad-icate fire blight and it is likely to spread to more orchards, resulting in severe situation (Choi et al., 2022b). Commercial products, including 12 antibiotics and 8 copper compounds, either in single form or as mixtures have been registered by the RDA as commercial control agents against fire blight. The copper and antibiotic products are recommended for treating orchards in the winter and blooming seasons, respectively. However, these products are not preferred by cultivators owing to their low efficacy, at least in field conditions, their side effects such as inadequate fertilization and abscission of immature fruits, and public concerns over their use. Thus, BCAs should be considered as alternative control agents against fire blight in Korea. The increasing use of BCAs every year is evident in the total year end sale. For example, the annual sales of Serenade from 2014 to 2018 increased by 331, 338, 35, 792, and 1,561 million Korean Won, respectively. In 2021, the annual sales of Serenade were almost two-fold higher than that in 2018, and it is predicted to further expand in the future (personal communication with Dr. Oh, K. H. in Farmhannong Ltd.).
Bacillus spp. have the benefits ease of growth, low toxicity to plants, and production of long-living spores (Whipps and McQuiken, 2009). The five BCAs selected and identified as Bacillus spp. in this study may also have the aforementioned benefits. The strains KPB 15 and 39 were identified as B. amyloliquefaciens and B. subtilis, respectively (Fig. 2), corresponding to previously identified BCAs against fire blight (Aldwinckle et al., 2002; Bahadou et al., 2018; Broggini et al., 2005; Chen et al., 2009; Shemshura et al., 2020). In addition, KPB 21, 25, and 31, identified as B. stratosphericus, B. altitudinis, and B. safensis, respectively, demonstrate antagonism against E. amylovora in this study. To date, these three Bacillus species have been reported as plant biocontrol agents. For example, B. stratosphericus, which was isolated from zinc-contaminat-ed mine soils, showed inhibition against five plant pathogenic bacteria, Burkholderia glumae, Xanthomonas oryzae pv. oryzae, Pectobacterium carotovorum, Pseudomonas syringae, and Ralstonia solanacearum by catalyzing siderophore production in vitro (Durairaj et al., 2017). In addition, B. altitudinis and B. safensis have been suggested as potential plant growth promoting rhizobacteria and suppression of fungal diseases (Sunar et al., 2015), and can be used as biopesticides to control rice blast and oomycetes plant pathogens (Bibi et al., 2012; Rong et al., 2020), respectively. Among the isolated BCAs, the antagonism test showed that KPB 39 exhibited antagonism against KPB 21, 25, and 31, indicating B. subtilis as the dominant species in the soil and that several commercial biopesticides containing B. subtilis should not be mixed with other biopesticides.
The biocontrol efficacy of the five selected BCAs varied in both immature apple fruits and seedlings, but their abilities to reduce disease were more consistent in seedlings than that in immature fruits (Fig. 3A, B). In a previous report, several Bacillus species were identified as endophytes (Bibi et al., 2012), suggesting that the five selected BCAs may also be endophytes as they occupy the inner tissues of seedlings with more ease than in immature fruits. However, in the present study, we did not evaluate whether these BCAs are endophytes, and did not identify the active ingredients related to their control effects against fire blight. Therefore, future studies should analyze the active ingredients such as antibiotics, enzymes, and peptides, via genetic analyses to elucidate the mechanism underlying their antagonism against E. amylovora. Nevertheless, KPB 25 and 31, individually and in a cocktail, showed the greatest reduction in lesion size on immature fruits and consistent effect in seedlings with no antagonism against the other four BCAs, and were finally chosen as novel single and mixed cocktail BCAs. These strains show promising action against E. amylovora and can be substitutes to imported BCAs, such as Serenade and Serifel.
Notes
Conflicts of Interest
Duck Hwan Park, a contributing editor of the Research in Plant Disease, was not involved in the editorial evaluation or decision to publish this article. All remaining authors have declared no conflicts of interest.
Acknowledgments
This work was carried out with the support of Korea Institute of Planning and Evaluation for Technology in Food, Agriculture, Forestry (IPET) through Agri-Bio industry Technology Development Program, funded by Ministry of Agriculture, Food and Rural Affairs (MAFRA) (No. 320041-05-2-SB010).