Ahlquist, P., Noueiry, A. O., Lee, W. M., Kushner, D. B. and Dye, B. T. 2003. Host factors in positive-strand RNA virus genome replication.
J. Virol 77: 8181-8186.



Ali, Z., Abul-Faraj, A., Piatek, M. and Mahfouz, M. M. 2015. Activity and specificity of TRV-mediated gene editing in plants.
Plant Signal Behav 10: e1044191



Amari, K., Boutant, E., Hofmann, C., Schmitt-Keichinger, C., Fernandez-Calvino, L., Didier, P. et al. 2010. A family of plasmodesmal proteins with receptor-like properties for plant viral movement proteins.
PLoS Pathog 6: e1001119



Amari, K., Di Donato, M., Dolja, V. V. and Heinlein, M. 2014. Myosins VIII and XI play distinct roles in reproduction and transport of tobacco mosaic virus.
PLoS Pathog 10: e1004448



Annamalai, P. and Rao, A. L. 2006. Delivery and expression of functional viral RNA genomes in planta by agroinfiltration.
Curr. Protoc. Microbiol 16: B.2.1-B.2.15.

Aouida, M., Eid, A., Ali, Z., Cradick, T., Lee, C., Deshmukh, H. et al. 2015a. Efficient fdCas9 synthetic endonuclease with improved specificity for precise genome engineering.
PLoS One 10: e0133373


Aouida, M., Piatek, M. J., Bangarusamy, D. K. and Mahfouz, M. M. 2014. Activities and specificities of homodimeric TALENs in Saccharomyces cerevisiae.
Curr. Genet 60: 61-74.


Aouida, M., Li, L., Mahjoub, A., Alshareef, S., Ali, Z., Piatek, A. et al. 2015b. Transcription activator-like effector nucleases mediated metabolic engineering for enhanced fatty acids production in Saccharomyces cerevisiae.
J. Biosci. Bioeng 120: 364-371.


Ashby, J., Boutant, E., Seemanpillai, M., Groner, A., Sambade, A., Ritzenthaler, C. et al. 2006. Tobacco mosaic virus movement protein functions as a structural microtubule-associated protein.
J. Virol 80: 8329-8344.



Barakate, A. and Stephens, J. 2016. An overview of CRISPR-based tools and their improvements:New opportunities in understanding plant-pathogen interactions for better crop protection.
Front. Plant Sci 7: 765



Barrangou, R. and Marraffini, L. A. 2014. CRISPR-Cas systems:Pro-karyotes upgrade to adaptive immunity.
Mol. Cell 54: 234-244.



Beauchemin, C., Boutet, N. and Laliberte, J. F. 2007. Visualization of the interaction between the precursors of VPg, the viral protein linked to the genome of turnip mosaic virus, and the translation eukaryotic initiation factor iso 4E in Planta.
J. Virol 81: 775-782.


Belov, G. A. and van Kuppeveld, F. J. 2012. (+)RNA viruses rewire cellular pathways to build replication organelles.
Curr. Opin. Virol 2: 740-747.



Blanc, S., Lopez-Moya, J. J., Wang, R., Garcia-Lampasona, S., Thornbury, D. W. and Pirone, T. P. 1997. A specific interaction between coat protein and helper component correlates with aphid transmission of a potyvirus.
Virology 231: 141-147.


Carrasco, J. L., Ancillo, G., Castello, M. J. and Vera, P. 2005. A novel DNA-binding motif, hallmark of a new family of plant transcription factors.
Plant Physiol 137: 602-606.



Castello, M. J., Carrasco, J. L. and Vera, P. 2010. DNA-binding protein phosphatase AtDBP1 mediates susceptibility to two potyviruses in Arabidopsis.
Plant Physiol 153: 1521-1525.



Cavatorta, J., Perez, K. W., Gray, S. M., Van Eck, J., Yeam, I. and Jahn, M. 2011. Engineering virus resistance using a modified potato gene.
Plant Biotechnol. J 9: 1014-1021.


Cermak, T., Curtin, S. J., Gil-Humanes, J., Cegan, R., Kono, T. J. Y., Konecna, E. et al. 2017. A multipurpose toolkit to enable advanced genome engineering in plants.
Plant Cell 29: 1196-1217.



Chandrasekaran, J., Brumin, M., Wolf, D., Leibman, D., Klap, C., Pearlsman, M. et al. 2016. Development of broad virus resistance in non-transgenic cucumber using CRISPR/Cas9 technology.
Mol. Plant Pathol 17: 1140-1153.



Cheng, X., Li, F., Cai, J., Chen, W., Zhao, N., Sun, Y. et al. 2015. Artificial TALE as a convenient protein platform for engineering broad-spectrum resistance to begomoviruses.
Viruses 7: 4772-4782.



Choi, S. H., Nakahara, K. S., Andrade, M. and Uyeda, I. 2012. Characterization of the recessive resistance gene
cyv1 of
Pisum sativum against
Clover yellow vein virus.
J. Gen. Plant Pathol 78: 269-276.

Clement, M., Leonhardt, N., Droillard, M. J., Reiter, I., Montillet, J. L., Genty, B. et al. 2011. The cytosolic/nuclear HSC70 and HSP90 molecular chaperones are important for stomatal closure and modulate abscisic acid-dependent physiological responses in Arabidopsis.
Plant Physiol 156: 1481-1492.



Curtin, S. J., Zhang, F., Sander, J. D., Haun, W. J., Starker, C., Baltes, N. J. et al. 2011. Targeted mutagenesis of duplicated genes in soybean with zinc-finger nucleases.
Plant Physiol 156: 466-473.



de Castro, I.F., Volonte, L. and Risco, C. 2013. Virus factories:biogenesis and structural design.
Cell Microbiol 15: 24-34.


den Boon, J.A., Diaz, A. and Ahlquist, P. 2010. Cytoplasmic viral replication complexes.
Cell Host Microbe 8: 77-85.



Diaz, A. and Wang, X. 2014. Bromovirus-induced remodeling of host membranes during viral RNA replication.
Curr. Opin. Virol 9: 104-110.


Diaz-Pendon, J. A., Truniger, V., Nieto, C., Garcia-Mas, J., Bendahmane, A. and Aranda, M. A. 2004. Advances in understanding recessive resistance to plant viruses.
Mol. Plant Pathol 5: 223-233.


Diez, J., Ishikawa, M., Kaido, M. and Ahlquist, P. 2000. Identification and characterization of a host protein required for efficient template selection in viral RNA replication.
Proc. Natl. Acad. Sci. U. S. A 97: 3913-3918.



Doudna, J. A. and Charpentier, E. 2014. Genome editing. The new frontier of genome engineering with CRISPR-Cas9.
Science 346: 1258096


Dunoyer, P., Thomas, C., Harrison, S., Revers, F. and Maule, A. 2004. A cysteine-rich plant protein potentiates Potyvirus movement through an interaction with the virus genome-linked protein VPg.
J. Virol 78: 2301-2309.



Feng, Z., Xue, F., Xu, M., Chen, X., Zhao, W., Garcia-Murria, M.J. et al. 2016. The ER-membrane transport system is critical for intercellular trafficking of the NSm movement protein and tomato spotted wilt tospovirus.
PLoS Pathog 12: e1005443



Fraser, R. S. S. 1990. The genetics of resistance to plant viruses.
Annu. Rev. Phytopathol 28: 179-200.

Gancarz, B. L., Hao, L., He, Q., Newton, M. A. and Ahlquist, P. 2011. Systematic identification of novel, essential host genes affecting bromovirus RNA replication.
PLoS One 6: e23988



Gao, Z., Johansen, E., Eyers, S., Thomas, C. L., Noel Ellis, T. H. and Maule, A. J. 2004. The potyvirus recessive resistance gene, sbm1, identifies a novel role for translation initiation factor eIF4E in cell-to-cell trafficking.
Plant J 40: 376-385.


Gazo, B. M., Murphy, P., Gatchel, J. R. and Browning, K. S. 2004. A novel interaction of Cap-binding protein complexes eukary-otic initiation factor (eIF) 4F and eIF(iso)4F with a region in the 3'-untranslated region of satellite tobacco necrosis virus.
J. Biol. Chem 279: 13584-13592.


Giner, A., Pascual, L., Bourgeois, M., Gyetvai, G., Rios, P., Pico, B. et al. 2017. A mutation in the melon vacuolar protein sorting 41prevents systemic infection of cucumber mosaic virus.
Sci. Rep 7: 10471



Hart, J. P. and Griffiths, P. D. 2013. A series of eIF4E alleles at the Bc-3 locus are associated with recessive resistance to clover yellow vein virus in common bean.
Theor. Appl. Genet 126: 2849-2863.


Hashimoto, M., Neriya, Y., Yamaji, Y. and Namba, S. 2016a. Recessive resistance to plant viruses:potential resistance genes beyond translation initiation factors.
Front. Microbiol 7: 1695


Hashimoto, M., Neriya, Y., Keima, T., Iwabuchi, N., Koinuma, H., Hagiwara-Komoda, Y. et al. 2016b. EXA1, a GYF domain protein, is responsible for loss-of-susceptibility to plantago asiatica mosaic virus in Arabidopsis thaliana.
Plant J 88: 120-131.


Heinlein, M. 2015. Plasmodesmata:channels for viruses on the move.
Methods Mol. Biol 1217: 25-52.


Hilliker, A., Gao, Z., Jankowsky, E. and Parker, R. 2011. The DEAD-box protein Ded1 modulates translation by the formation and resolution of an eIF4F-mRNA complex.
Mol. Cell 43: 962-972.



Ho, Y., Gruhler, A., Heilbut, A., Bader, G. D., Moore, L., Adams, S. L. et al. 2002. Systematic identification of protein complexes in Saccharomyces cerevisiae by mass spectrometry.
Nature 415: 180-183.



Hofinger, B. J., Russell, J. R., Bass, C. G., Baldwin, T., dos Reis, M., Hedley, P. E. et al. 2011. An exceptionally high nucleotide and haplotype diversity and a signature of positive selection for the eIF4E resistance gene in barley are revealed by allele mining and phylogenetic analyses of natural populations.
Mol. Ecol 20: 3653-3668.


Ishikawa, M., Obata, F., Kumagai, T. and Ohno, T. 1991. Isolation of mutants of Arabidopsis thaliana in which accumulation of to-bacco mosaic virus coat protein is reduced to low levels.
Mol. Gen. Genet 230: 33-38.


Ishikawa, M., Naito, S. and Ohno, T. 1993. Effects of the tom1 mutation of Arabidopsis thaliana on the multiplication of tobacco mosaic virus RNA in protoplasts.
J. Virol 67: 5328-5338.



Jiang, J. and Laliberte, J. F. 2011. The genome-linked protein VPg of plant viruses-a protein with many partners.
Curr. Opin. Virol 1: 347-354.


Jiang, J., Patarroyo, C., Garcia Cabanillas, D., Zheng, H. and Laliberte, J. F. 2015. The vesicle-forming 6K2 protein of turnip mosaic virus interacts with the COPII coatomer Sec24a for viral systemic infection.
J. Virol 89: 6695-6710.



Julio, E., Cotucheau, J., Decorps, C., Volpatti, R., Sentenac, C., Candresse, T. et al. 2015. A eukaryotic translation initiation factor 4E (eIF4E) is responsible for the “
va”tobacco recessive resistance to potyviruses.
Plant Mol. Biol. Rep 33: 609-623.

Jungkunz, I., Link, K., Vogel, F., Voll, L. M., Sonnewald, S. and Son-newald, U. 2011. AtHsp70-15-deficient Arabidopsis plants are characterized by reduced growth, a constitutive cytosolic protein response and enhanced resistance to TuMV.
Plant J 66: 983-995.


Kang, B. C., Yeam, I. and Jahn, M. M. 2005. Genetics of plant virus resistance.
Annu. Rev. Phytopathol 43: 581-621.


Kang, B. C., Yeam, I., Li, H., Perez, K. W. and Jahn, M. M. 2007. Ectopic expression of a recessive resistance gene generates dominant potyvirus resistance in plants.
Plant Biotechnol. J 5: 526-536.


Keen, N. T. 1990. Gene-for-gene complementarity in plant-pathogen interactions.
Annu. Rev. Genet 24: 447-463.


Kumar, S., Dubey, A. K., Karmakar, R., Kini, K. R., Mathew, M. K. and Prakash, H. S. 2012. Inhibition of TMV multiplication by siRNA constructs against TOM1 and TOM3 genes of Capsicum annuum.
J. Virol. Methods 186: 78-85.


Kushner, D. B., Lindenbach, B. D., Grdzelishvili, V. Z., Noueiry, A. O., Paul, S. M. and Ahlquist, P. 2003. Systematic, genome-wide identification of host genes affecting replication of a positive-strand RNA virus.
Proc. Natl. Acad. Sci. U. S. A 100: 15764-15769.



Langner, T., Kamoun, S. and Belhaj, K. 2018. CRISPR crops: Plant genome editing toward disease resistance.
Annu. Rev. Phytopathol 56: 479-512.


Lee, M. W. and Yang, Y. 2006. Transient expression assay by agroinfiltration of leaves.
Methods Mol. Biol 323: 225-229.


Legg, J. P., Shirima, R., Tajebe, L. S., Guastella, D., Boniface, S., Jeremiah, S. et al. 2014. Biology and management of Bemisia whitefly vectors of cassava virus pandemics in Africa.
Pest Manag. Sci 70: 1446-1453.


Lellis, A. D., Kasschau, K. D., Whitham, S. A. and Carrington, J. C. 2002. Loss-of-susceptibility mutants of Arabidopsis thaliana reveal an essential role for eIF(iso)4E during potyvirus infection.
Curr. Biol 12: 1046-1051.


Lewis, J. D. and Lazarowitz, S. G. 2010. Arabidopsis synaptotagmin SYTA regulates endocytosis and virus movement protein cell-to-cell transport.
Proc. Natl. Acad. Sci. U. S. A 107: 2491-2496.



Lin, J. W., Ding, M. P., Hsu, Y. H. and Tsai, C. H. 2007. Chloroplast phosphoglycerate kinase, a gluconeogenetic enzyme, is required for efficient accumulation of Bamboo mosaic virus.
Nucleic Acids Res 35: 424-432.


Loebenstein, G. and Katis, N. 2014. Control of plant virus diseases seed-propagated crops. Preface.
Adv. Virus Res 90: xi

Maia, I. G., Haenni, A. and Bernardi, F. 1996. Potyviral HC-Pro: a multifunctional protein.
J. Gen. Virol 77: 1335-1341.


Mann, M., Hendrickson, R. C. and Pandey, A. 2001. Analysis of proteins and proteomes by mass spectrometry.
Annu. Rev. Biochem 70: 437-473.


Martinez-Silva, A. V., Aguirre-Martinez, C., Flores-Tinoco, C. E., Ale-jandri-Ramirez, N. D. and Dinkova, T. D. 2012. Translation initiation factor AteIF(iso)4E is involved in selective mRNA translation in Arabidopsis thaliana seedlings.
PLoS One 7: e31606



Maule, A. J. 2008. Plasmodesmata:structure, function and biogenesis.
Curr. Opin. Plant Biol 11: 680-686.


Mayberry, L. K., Allen, M. L., Nitka, K. R., Campbell, L., Murphy, P. A. and Browning, K. S. 2011. Plant cap-binding complexes eukary-otic initiation factors eIF4F and eIFISO4F:molecular specificity of subunit binding.
J. Biol. Chem 286: 42566-42574.



Nagy, P. D. 2016. Tombusvirus-Host interactions:Co-Opted evolutionarily conserved host factors take center court.
Annu. Rev. Virol 3: 491-515.


Nagy, P. D. and Pogany, J. 2011. The dependence of viral RNA replication on co-opted host factors.
Nat. Rev. Microbiol 10: 137-149.



Nagy, P. D. and Richardson, C. D. 2012. Viral replication-in search of the perfect host.
Curr. Opin. Virol 2: 663-668.


Nicaise, V., German-Retana, S., Sanjuan, R., Dubrana, M. P., Mazier, M., Maisonneuve, B. et al. 2003. The eukaryotic translation initiation factor 4E controls lettuce susceptibility to the Potyvirus Lettuce mosaic virus.
Plant Physiol 132: 1272-1282.



Nicaise, V., Gallois, J. L., Chafiai, F., Allen, L. M., Schurdi-Levraud, V., Browning, K. S. et al. 2007. Coordinated and selective recruitment of eIF4E and eIF4G factors for potyvirus infection in Arabi-dopsis thaliana.
FEBS Lett 581: 1041-1046.


Nieto, C., Morales, M., Orjeda, G., Clepet, C., Monfort, A., Sturbois, B. et al. 2006. An eIF4E allele confers resistance to an uncapped and non-polyadenylated RNA virus in melon.
Plant J 48: 452-462.


Nishikiori, M., Mori, M., Dohi, K., Okamura, H., Katoh, E., Naito, S. et al. 2011. A host small GTP-binding protein ARL8 plays crucial roles in tobamovirus RNA replication.
PLoS Pathog 7: e1002409



Noueiry, A. O., Chen, J. and Ahlquist, P. 2000. A mutant allele of essential, general translation initiation factor DED1 selectively inhibits translation of a viral mRNA.
Proc. Natl. Acad. Sci. U. S. A 97: 12985-12990.



Noueiry, A. O., Diez, J., Falk, S. P., Chen, J. and Ahlquist, P. 2003. Yeast Lsm1p-7p/Pat1p deadenylation-dependent mRNA-decapping factors are required for brome mosaic virus genomic RNA translation.
Mol. Cell Biol 23: 4094-4106.



Ohshima, K., Taniyama, T., Yamanaka, T., Ishikawa, M. and Naito, S. 1998. Isolation of a mutant of Arabidopsis thaliana carrying two simultaneous mutations affecting tobacco mosaic virus multi-plication within a single cell.
Virology 243: 472-481.


Orjuela, J., Deless, E. F., Kolade, O., Cheron, S., Ghesquiere, A. and Albar, L. 2013. A recessive resistance to rice yellow mottle virus is associated with a rice homolog of the CPR5 gene, a regulator of active defense mechanisms.
Mol. Plant-Microbe Interact 26: 1455-1463.


Ouibrahim, L., Mazier, M., Estevan, J., Pagny, G., Decroocq, V., Desbiez, C. et al. 2014. Cloning of the Arabidopsis rwm1 gene for resistance to Watermelon mosaic virus points to a new function for natural virus resistance genes.
Plant J 79: 705-716.


Ouko, M. O., Sambade, A., Brandner, K., Niehl, A., Pena, E., Ahad, A. et al. 2010. Tobacco mutants with reduced microtubule dynamics are less susceptible to TMV.
Plant J 62: 829-839.


Patrick, R. M., Mayberry, L. K., Choy, G., Woodard, L. E., Liu, J. S., White, A. et al. 2014. Two Arabidopsis loci encode novel eukary-otic initiation factor 4E isoforms that are functionally distinct from the conserved plant eukaryotic initiation factor 4E.
Plant Physiol 164: 1820-1830.



Perez, K., Yeam, I., Kang, B. C., Ripoll, D. R., Kim, J., Murphy, J. F. et al. 2012. Tobacco etch virus infectivity in Capsicum spp. is determined by a maximum of three amino acids in the viral virulence determinant VPg.
Mol. Plant-Microbe Interact 25: 1562-1573.


Piatek, A. and Mahfouz, M. M. 2017. Targeted genome regulation via synthetic programmable transcriptional regulators.
Crit. Rev. Biotechnol 37: 429-440.


Poque, S., Pagny, G., Ouibrahim, L., Chague, A., Eyquard, J. P., Caballero, M. et al. 2015. Allelic variation at the rpv1 locus controls partial resistance to Plum pox virus infection in Arabidopsis thaliana.
BMC Plant Biol 15: 159



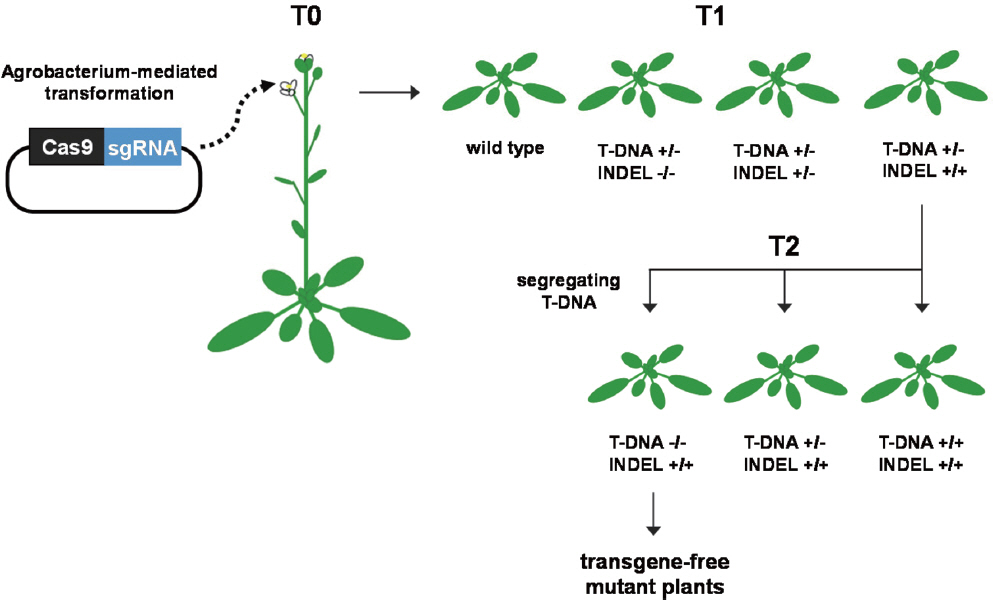
Pyott, D. E., Sheehan, E. and Molnar, A. 2016. Engineering of CRISPR/Cas9-mediated potyvirus resistance in transgene-free Arabi-dopsis plants.
Mol. Plant Pathol 17: 1276-1288.



Quetier, F. 2016. The CRISPR-Cas9 technology: Closer to the ultimate toolkit for targeted genome editing.
Plant Sci 242: 65-76.


Ransom-Hodgkins, W. D. 2009. The application of expression analysis in elucidating the eukaryotic elongation factor one alpha gene family in Arabidopsis thaliana.
Mol. Genet. Genomics 281: 391-405.


Robaglia, C. and Caranta, C. 2006. Translation initiation factors: a weak link in plant RNA virus infection.
Trends Plant Sci 11: 40-45.


Roudet-Tavert, G., German-Retana, S., Delaunay, T., Delecolle, B., Candresse, T. and Le Gall, O. 2002. Interaction between potyvirus helper component-proteinase and capsid protein in infected plants.
J. Gen. Virol 83: 1765-1770.


Roudet-Tavert, G., Michon, T., Walter, J., Delaunay, T., Redondo, E. and Le Gall, O. 2007. Central domain of a potyvirus VPg is involved in the interaction with the host translation initiation factor eIF4E and the viral protein HcPro.
J. Gen. Virol 88: 1029-1033.


Ruffel, S., Dussault, M. H., Palloix, A., Moury, B., Bendahmane, A., Robaglia, C. et al. 2002. A natural recessive resistance gene against potato virus Y in pepper corresponds to the eukaryotic initiation factor 4E (eIF4E).
Plant J 32: 1067-1075.


Ruffel, S., Gallois, J. L., Lesage, M. L. and Caranta, C. 2005. The recessive potyvirus resistance gene pot-1 is the tomato orthologue of the pepper pvr2-eIF4E gene.
Mol. Genet. Genomics 274: 346-353.


Ruffel, S., Gallois, J. L., Moury, B., Robaglia, C., Palloix, A. and Caranta, C. 2006. Simultaneous mutations in translation initiation factors eIF4E and eIF(iso)4E are required to prevent pepper veinal mottle virus infection of pepper.
J. Gen. Virol 87: 2089-2098.


Sambade, A., Brandner, K., Hofmann, C., Seemanpillai, M., Mutterer, J. and Heinlein, M. 2008. Transport of TMV movement protein particles associated with the targeting of RNA to plasmodesmata.
Traffic 9: 2073-2088.


Sato, M., Nakahara, K., Yoshii, M., Ishikawa, M. and Uyeda, I. 2005. Selective involvement of members of the eukaryotic initiation factor 4E family in the infection of Arabidopsis thaliana by potyviruses.
FEBS Lett 579: 1167-1171.


Seo, J. K., Choi, H. S. and Kim, K. H. 2016. Engineering of soybean mosaic virus as a versatile tool for studying protein-protein interactions in soybean.
Sci. Rep 6: 22436



Stella, S. and Montoya, G. 2016. The genome editing revolution:A CRISPR-Cas TALE off-target story.
Bioessays 38: S4-S13.


Truniger, V. and Aranda, M. A. 2009. Recessive resistance to plant viruses.
Adv. Virus Res 75: 119-159.


Tsujimoto, Y., Numaga, T., Ohshima, K., Yano, M. A., Ohsawa, R., Goto, D. B. et al. 2003. Arabidopsis TOBAMOVIRUS MULTIPLICATION (TOM) 2 locus encodes a transmembrane protein that interacts with TOM1.
EMBO J 22: 335-343.



Uchiyama, A., Shimada-Beltran, H., Levy, A., Zheng, J. Y., Javia, P. A. and Lazarowitz, S. G. 2014. The Arabidopsis synaptotagmin SYTA regulates the cell-to-cell movement of diverse plant viruses.
Front. Plant Sci 5: 584



Vaghchhipawala, Z., Rojas, C. M., Senthil-Kumar, M. and Mysore, K. S. 2011. Agroinoculation and agroinfiltration:simple tools for complex gene function analyses.
Methods Mol. Biol 678: 65-76.


Vasilescu, J. and Figeys, D. 2006. Mapping protein-protein interactions by mass spectrometry.
Curr. Opin. Biotechnol 17: 394-399.


Vijayapalani, P., Maeshima, M., Nagasaki-Takekuchi, N. and Miller, W. A. 2012. Interaction of the trans-frame potyvirus protein P3N-PIPO with host protein PCaP1 facilitates potyvirus movement.
PLoS Pathog 8: e1002639



Wang, A. 2015. Dissecting the molecular network of virus-plant interactions:the complex roles of host factors.
Annu. Rev. Phytopathol 53: 45-66.


Wang, A. and Krishnaswamy, S. 2012. Eukaryotic translation initiation factor 4E-mediated recessive resistance to plant viruses and its utility in crop improvement.
Mol. Plant Pathol 13: 795-803.



Wang, Y., Cheng, X., Shan, Q., Zhang, Y., Liu, J., Gao, C. et al. 2014. Simultaneous editing of three homoeoalleles in hexaploid bread wheat confers heritable resistance to powdery mildew.
Nat. Biotechnol 32: 947-951.


Whitham, S. A., Yamamoto, M. L. and Carrington, J. C. 1999. Selectable viruses and altered susceptibility mutants in Arabidopsis thaliana.
Proc. Natl. Acad. Sci. U. S. A 96: 772-777.



Wittmann, S., Chatel, H., Fortin, M. G. and Laliberte, J. F. 1997. Interaction of the viral protein genome linked of turnip mosaic potyvirus with the translational eukaryotic initiation factor (iso) 4E of Arabidopsis thaliana using the yeast two-hybrid system.
Virology 234: 84-92.


Wright, A. V., Nunez, J. K. and Doudna, J. A. 2016. Biology and applications of CRISPR systems:Harnessing nature's toolbox for genome engineering.
Cell 164: 29-44.


Yamanaka, T., Imai, T., Satoh, R., Kawashima, A., Takahashi, M., Tomita, K. et al. 2002. Complete inhibition of tobamovirus multiplication by simultaneous mutations in two homologous host genes.
J. Virol 76: 2491-2497.



Yamanaka, T., Ohta, T., Takahashi, M., Meshi, T., Schmidt, R., Dean, C. et al. 2000. TOM1, an Arabidopsis gene required for efficient multiplication of a tobamovirus, encodes a putative transmembrane protein.
Proc. Natl. Acad. Sci. U. S. A 97: 10107-10112.



Yang, P., Perovic, D., Habekuß, A., Zhou, R., Graner, A., Ordon, F. et al. 2013. Gene-based high-density mapping of the gene
rym7 conferring resistance to
Barley mild mosaic virus(BaMMV).
Mol. Breed 32: 27-37.

Yang, P., Lupken, T., Habekuss, A., Hensel, G., Steuernagel, B., Kilian, B. et al. 2014. PROTEIN DISULFIDE ISOMERASE LIKE 5-1 is a sus-ceptibility factor to plant viruses.
Proc. Natl. Acad. Sci. U. S. A 111: 2104-2109.



Ye, C., Dickman, M. B., Whitham, S. A., Payton, M. and Verchot, J. 2011. The unfolded protein response is triggered by a plant viral movement protein.
Plant Physiol 156: 741-755.



Yoshii, M., Nishikiori, M., Tomita, K., Yoshioka, N., Kozuka, R., Naito, S. et al. 2004. The Arabidopsis cucumovirus multiplication 1 and 2 loci encode translation initiation factors 4E and 4G.
J. Virol 78: 6102-6111.



Yoshii, M., Shimizu, T., Yamazaki, M., Higashi, T., Miyao, A., Hirochika, H. et al. 2009. Disruption of a novel gene for a NAC-domain protein in rice confers resistance to rice dwarf virus.
Plant J 57: 615-625.


Yoshii, M., Yoshioka, N., Ishikawa, M. and Naito, S. 1998a. Isolation of an Arabidopsis thaliana mutant in which accumulation of cucumber mosaic virus coat protein is delayed.
Plant J 13: 211-219.


Yoshii, M., Yoshioka, N., Ishikawa, M. and Naito, S. 1998b. Isolation of an Arabidopsis thaliana mutant in which the multiplication of both cucumber mosaic virus and turnip crinkle virus is affected.
J. Virol 72: 8731-8737.


Zetsche, B., Gootenberg, J. S., Abudayyeh, O. O., Slaymaker, I. M., Makarova, K. S., Essletzbichler, P. et al. 2015. Cpf1 is a single RNA-guided endonuclease of a class 2 CRISPR-Cas system.
Cell 163: 759-771.



Zhang, L., Chen, H., Brandizzi, F., Verchot, J. and Wang, A. 2015a. The UPR branch IRE1-bZIP60 in plants plays an essential role in viral infection and is complementary to the only UPR pathway in yeast.
PLoS Genet 11: e1005164


Zhang, X. C., Millet, Y. A., Cheng, Z., Bush, J. and Ausubel, F. M. 2015b. Jasmonate signalling in Arabidopsis involves SGT1b-HSP70-HSP90 chaperone complexes.
Nat. Plants 1: 15049


Zong, Y., Wang, Y., Li, C., Zhang, R., Chen, K., Ran, Y. et al. 2017. Precise base editing in rice, wheat and maize with a Cas9-cytidine deaminase fusion.
Nat. Biotechnol 35: 438-440.


Zou, L. J., Deng, X. G., Han, X. Y., Tan, W. R., Zhu, L. J., Xi, D. H. et al. 2016. Role of transcription factor HAT1 in modulating arabi-dopsis thaliana response to cucumber mosaic virus.
Plant Cell Physiol 57: 1879-1889.







 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print