Geiser, D. M, del Mar Jiménez-Gasco, M, Kang, S, Makalowska, I, Veeraraghavan, N, Ward, T. J, Zhang, N, Kuldau, G. A and O’donnell, K FUSARIUM-ID v. 1.0: A DNA sequence database for identifying Fusarium.
Eur. J. Plant Pathol 2004. 110: 473-479.

Han, D. S Sangyakhak. 1988. Seoul, Korea. Dongmyungsa Press, (In Korean)
Hsuan, H. M, Salleh, B and Zakaria, L Molecular identification of
Fusarium species in
Gibberella fujikuroi species complex from rice, sugarcane and maize from Peninsular Malaysia.
Int. J. Mol. Sci 2011. 12: 6722-6732.



Ichikawa, K and Takayuki, A New leaf spot disease of
Cymbidium species caused by
Fusarium subglutinans and
Fusarium proliferatum.
J. Gen. Plant Pathol 2000. 66: 213-218.


Kang, G. H, Chang, E. J and Choi, S. W Antioxidative activity of phenolic compounds in roasted safflower (Carthamus tinctorius L.) seeds. J. Food Sci. Nutr 1999. 4: 221-225.
Kim, I. H Sinyakboncho. 1992. Seoul, Korea. Insandongcheon Press, (In Korean)
Lee, C. B Picture Book of Korean Plants. 1980. Seoul, Korea. Baekyang Press, (In Korean)
Lee, J. Y, Chang, E. J, Kim, H. J, Park, J. H and Choi, S. W Antioxidative flavonoids from leaves of
Carthamus tinctorius.
Arch. Pharm. Res 2002. 25: 313-319.



Nelson, P. E, Toussoun, T. A and Marasas, W. F. O Fusarium Species: An Illustrated Manual for Identification. 1983. University Park, PA, USA. Pennsylvania State University Press.
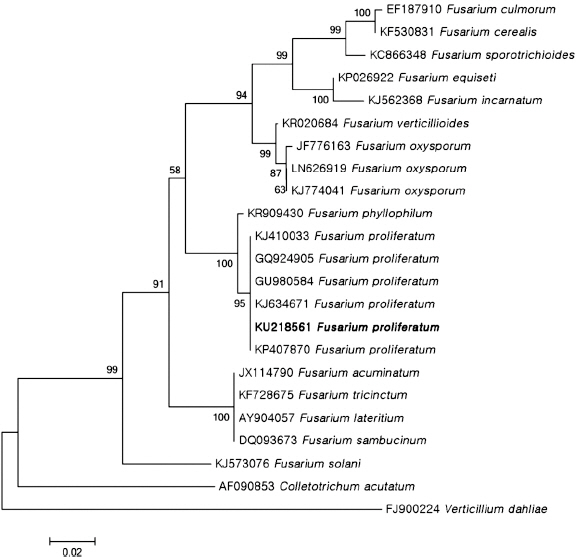
Saitou, N and Nei, M The neighbor-joining method: a new method for reconstructing phylogenetic trees.
Mol. Biol. Evol 1987. 4: 406-425.

Tamura, K, Stecher, G, Peterson, D, Filipski, A and Kumar, S MEGA6: molecular evolutionary genetics analysis version 6.0.
Mol. Biol. Evol 2013. 30: 2725-2729.



White, T. J, Bruns, T, Lee, S and Taylor, J. W eds. by M. A Innis, D. H Gelfand, J. J Sninsky and T. J White, Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: PCR Protocols: A Guide to Methods and Applications, 1990. pp. 315-322. New York, NY, USA. Academic Press Inc,




 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print