Bartz, J. A. 2003. Alternaria rot. Compendium of Pepper Diseases K. Pernezny, P. D. Roberts, J. F. Murphy and N. P. Goldberg 42-43. APS Press; St. Paul, MN, USA.
Berbee, M. L., Pirseyedi, M. and Hubbard, S. 1999.
Cochliobolus phylogenetics and the origin of known, highly virulent pathogens, inferred from ITS and glyceraldehyde-3-phosphate dehydrogenase gene sequences.
Mycologia 91: 964-977.

Carbone, I. and Kohn, L. M. 1999. A method for designing primer sets for speciation studies in filamentous ascomycetes.
Mycologia 91: 553-556.

Davis, R. M. and Paulus, A. O. 2014. Black mold. In: Compendium of Tomato Diseases and Pests, Second edition eds. by J. B. Jones, T. A. Zitter, T. M. Momol and S. A. Miller, pp. 18APS Press, St. Paul, MN, USA.
Dong, L., Liu, S., Li, J., Tharreau, D., Liu, P., Tao, D. et al. 2022. A rapid and simple method for DNA preparation of
Magnaporthe oryzae from single rice blast lesions for PCR-based molecular analysis.
Plant Pathol. J. 38: 679-684.




Edgar, R. C. 2004. MUSCLE: multiple sequence alignment with high accuracy and high throughput.
Nucleic. Acids Res. 32: 1792-1797.



Ellis, M. B. 1971. Dematiaceous Hyphomycetes. Commonwealth Mycological Institute, Kew, UK. pp. 608 pp.
Farr, D. F., Bills, G. F., Chamuris, G. P. and Rossman, A. Y. 1989. Fungi on Plants and Plant Products in the United States. APS Press, St. Paul, MN, USA. pp. 1252 pp.
Hong, S. G., Cramer, R. A., Lawrence, C. B. and Pryor, B. M. 2005. Alt a 1 allergen homologs from
Alternaria and related taxa: analysis of phylogenetic content and secondary structure.
Fungal Genet. Biol. 42: 119-129.


Kim, W. G., Ryu, J. T. and Choi, H. W. 2020. Black mold on tomato fruits caused by Alternaria alternata in Korea. Korean J. Mycol. 48: 369-379. (In Korean)
Kim, W. G. and Yu, S. H. 1985. A black mold of pepper fruits caused by. Alternaria alternata. Korean J. Plant Pathol. 1: 67-71. (In Korean)
Liu, Y. J., Whelen, S. and Hall, B. D. 1999. Phylogenetic relationships among ascomycetes: evidence from an RNA polymerse II subunit.
Mol. Biol. Evol. 16: 1799-1808.


Rotem, J. 1994. The Genus Alternaria, Biology, Epidemiology, and Pathogenicity. APS Press, St. Paul, MN, USA. pp. 326 pp.
Simmons, E. G. 2007.
Alternaria, An Identification Manual. CBS Fungal Biodiversity Centre, Utrecht, The Netherlands. pp. 775 pp.
Sung, G. H., Sung, J. M., Hywel-Jones, N. L. and Spatafora, J. W. 2007. A multi-gene phylogeny of Clavicipitaceae (Ascomycota, Fungi): identification of localized incongruence using a combinational bootstrap approach.
Mol. Phylogenet. Evol. 44: 1204-1223.


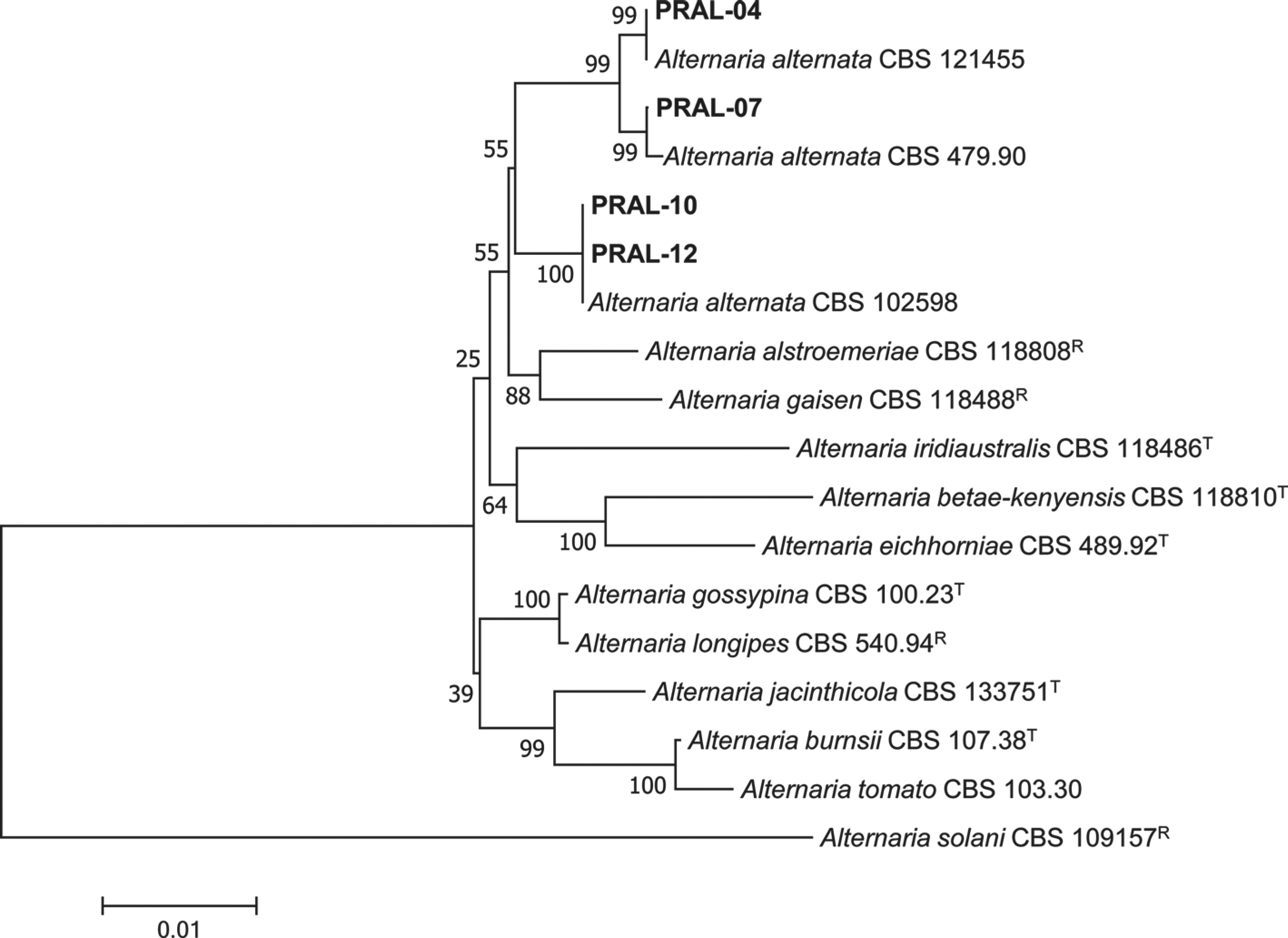
Woudenberg, J. H. C., Groenewald, J. Z., Binder, M. and Crous, P. W. 2013.
Alternaria redefined.
Stud. Mycol. 75: 171-212.



Woudenberg, J. H. C., Seidl, M. F., Groenewald, J. Z., De Vries, M., Stielow, J. B., Thomma, B. P. H. J. et al. 2015.
Alternaria section
Alternaria: species,
formae speciales or pathotypes?
Stud. Mycol. 82: 1-21.



Woudenberg, J. H. C., Truter, M., Groenewald, J. Z. and Crous, P. W. 2014. Large-spored
Alternaria pathogens in section
Porri disentangled.
Stud. Mycol. 79: 1-47.









 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print